BPC-157
BPC-157 promotes vascular endothelial growth factor (VEGF), aiding blood vessel formation and tissue repair. It counters growth-inhibitory effects of 4‐hydroxynonenal, boosts growth receptor activity, and enhances fibroblast production essential for collagen synthesis.
BPC 157
Common Name: BPC-157
Category: Stable Pentadecapeptide
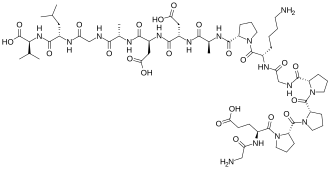
Chemical Structures:
BPC-157: Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val (15 amino acids)
Alternative Names: Body Protection Compound, PLD-116, PL-10, PL14736,Bepecin
CAS NUMBER: 137525-51-0
WHAT IS BPC 157?
BPC-157 is characterized as a stable gastric pentadecapeptide, meaning its structure is highly resistant to degradation, including in stomach acid. Its therapeutic potential stems from its pleiotropic (multifaceted) effects on cellular signaling pathways:
-
Angiogenesis and Vascular Repair: BPC-157 strongly promotes the formation of new blood vessels (angiogenesis). It primarily achieves this by activating the VEGFR2-Akt-eNOS signaling pathway, which enhances nitric oxide (NO) production, leading to vasodilation and improved vascular stability and perfusion. This collateral vessel formation is crucial for healing ischemic (low blood flow) injuries.
-
Fibroblast and Tendon Healing: For musculoskeletal repair, BPC-157 significantly accelerates the migration and proliferation of tendon fibroblasts, which are the cells responsible for producing collagen and repairing structural tissue. This action is mediated by the Focal Adhesion Kinase (FAK)-paxillin signaling pathway, which is critical for cell movement and survival.
-
Growth Factor Modulation: It upregulates growth hormone receptor (GHR) expression in target tissues like fibroblasts, which augments the tissue's anabolic (building) healing response. It also counters the growth-inhibitory effects of cellular toxins like 4-hydroxynonenal.
-
Cytoprotection: BPC-157 is widely studied for its ability to maintain and restore the integrity of the gastric and intestinal mucosa, providing protection against various damaging agents, including NSAIDs and alcohol.
-
Neurological Effects: Research suggests BPC-157 possesses neuroprotective properties and can modulate the dopaminergic and serotonergic systems, showing potential in models of traumatic brain injury and mood disorders.
WHAT DOES BPC-157 DO?
BPC-157 promotes vascular endothelial growth factor (VEGF), aiding blood vessel formation and tissue repair. It counters the growth-inhibitory effects of 4‐hydroxynonenal, boosts growth receptor activity, and enhances fibroblast production essential for collagen synthesis. In essence, it supercharges your body's natural repair function to see localized results in faster and more pronounced time periods.
BENEFITS & CLINICAL TRIALS
The experimental application of BPC-157 is extensive, largely supported by animal models:
| Research Focus | Experimental Benefit |
| Musculoskeletal Repair | Promotes faster healing from tendon, ligament, muscle injuries, bone fractures, and bone-to-muscle reattachment (e.g., re-establishing myotendinous junctions). |
| Gastrointestinal Health | Protects against ulcers, promotes the healing of inflammatory bowel disease models, and preserves the mucosal barrier. |
| Wound Healing | Accelerates wound closure, promotes epithelial regeneration, and reduces fibrosis (scar tissue formation). |
| Inflammation | Exerts powerful anti-inflammatory effects in both acute and chronic conditions. |
Clinical Observations (Limited Data)
While large-scale randomized controlled trials in humans are lacking, one retrospective human study noted that 7 out of 12 patients with chronic knee pain reported pain relief lasting for over six months after receiving a single intra-articular BPC-157 injection. Animal studies have generally shown an excellent safety profile, but these findings do not equate to established clinical safety data in humans.
SIDE EFFECTS
Given the lack of comprehensive human testing, the safety profile remains unknown. Anecdotal or preclinical side effects noted include:
- Injection site reactions (ISRs) are a common localised side effect that can occur following the administration of any injected vaccine. They are an inflammatory response to the injected vaccine. Symptoms of ISRs include swelling, redness (erythema), induration (hardness), pain or itch at or near the injection site.
- Peptide coupling agents help form amide bonds between a carboxylic acid and an amine, so the compounds have the potential to modify human proteins. This property makes the agents potential sensitizers, chemicals that can cause people who are regularly exposed to them to develop a potential allergy.
- The synthetically produced peptide BPC-157 is not currently approved for use as a human drug. It is an experimental compound that has been investigated for inflammatory bowel disease and soft tissue healing, although there is a concerning lack of published clinical trial data because studies appear to have been cancelled or stopped without any published conclusions.
IS BPC-157 SAFE?
CRITICAL WARNING
BPC-157 is an experimental, unapproved compound. Its safety profile has not been established through large-scale, independent human clinical trials required by global regulatory bodies like the FDA.
| Potential Risks and Unknowns | Detail |
| Lack of Regulation | The FDA has classified BPC-157 as a substance that should not be compounded, citing a lack of comprehensive safety data and potential risks for immunogenicity. |
| Long-Term Safety | The long-term effects of BPC-157 use in humans remain completely unknown. There is no data to assess its potential impact on cancer, tumor growth (given its cellular proliferation properties), or long-term effects on fertility. |
| Injection Risks | Common localized side effects include Injection Site Reactions (ISRs) such as swelling, pain, redness (erythema), or induration (hardness) at or near the injection site. |
| Chemical Sensitization | The synthetic nature of the peptide means that traces of peptide coupling agents used in manufacturing could pose a risk of acting as sensitizers, potentially inducing allergic reactions in individuals with regular exposure. |
Conclusion on Safety: While animal data suggests a benign profile, the use of BPC-157 for human health purposes is purely experimental and carries significant, unknown risks due to the absence of conclusive human safety data.
Administration
This information is derived from common practices in non-FDA regulated research and is for informational purposes only.
Reconstitution Procedure
-
Materials Needed: Vial of BPC-157 10mg lyophilized peptide powder, vial of Bacteriostatic Water (BAC Water), mixing syringe (e.g., 100-unit/1mL insulin syringe), and alcohol swabs.
-
Mixing: Inject the BAC Water slowly, directing the stream down the inside wall of the BPC-157 vial.
-
Peptide Preservation: DO NOT SHAKE the vial vigorously, as this can degrade the peptide structure. Instead, gently swirl the vial or roll it between your hands for several minutes until the powder is fully dissolved and the solution is clear.
-
Result: Adding 1.7mL of BAC water to a 10mg vial yields a concentration of 5.88mg per mL (0.588mg per 10 units on a U-100 syringe).
Dosage Regulations (Anecdotal Use)
-
Administration Method: Subcutaneous (SubQ) or Intramuscular (IM) injection is typically preferred for musculoskeletal issues, as it allows for localized administration near the injury site, maximizing bioavailability.
-
General Daily Range: 200mcg to 1,000mcg.
-
Common Standard Dose: 500mcg per day.
-
Localized Protocol (Tendons/Ligaments): 250–500mcg, once or twice daily. Administration should be as close as possible to the site of injury for optimal localized effects.
-
Cycle Length: Typically 2–4 weeks for minor injuries, extending up to 6 weeks for severe or post-operative recovery.
WHERE TO BUY BPC-157 STACK
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for BPC-157, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of BPC-157 outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
Team, Editorial. “BPC-157 Research Guide: The Science behind the Peptide.” BioLongevity Labs, 21 Oct. 2025, biolongevitylabs.com/research/bpc-157-research-guide/?tw_source=google&tw_adid=&tw_campaign=22124776050&tw_kwdid=&gad_source=1&gad_campaignid=22131035699&gbraid=0AAAAA-VNyo4Uc2QKR49_lCuPUVOX6zg2r&gclid=CjwKCAiA_dDIBhB6EiwAvzc1cKft0WRs6sQ0g4ZyatLURv1wDaztFft_acZ_F6dJo8AujaiD8dkVJhoCizUQAvD_BwE.
Vasireddi, Nikhil, et al. “Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review.” HSS Journal : The Musculoskeletal Journal of Hospital for Special Surgery, U.S. National Library of Medicine, 31 July 2025, pmc.ncbi.nlm.nih.gov/articles/PMC12313605/#:~:text=At%20this%20time%2C%20there%20is,at%20an%20all%2Dtime%20high.
“BPC-157.” Wikipedia, Wikimedia Foundation, 22 Oct. 2025, en.wikipedia.org/wiki/BPC-157#:~:text=Gastric%20Pentadecapeptide%20BPC%2D157%20(also,%2DGly%2DLeu%2DVal.
“What You Need to Know about BPC 157.” Healthy Male, healthymale.org.au/health-article/bpc157-side-effects.
Chang CH;Tsai WC;Lin MS;Hsu YH;Pang JH; “The Promoting Effect of Pentadecapeptide BPC 157 on Tendon Healing Involves Tendon Outgrowth, Cell Survival, and Cell Migration.” Journal of Applied Physiology (Bethesda, Md. : 1985), U.S. National Library of Medicine, pubmed.ncbi.nlm.nih.gov/21030672/.
Boerner, Leigh Krietsch. “Peptide Coupling Agents Can Cause Severe Allergic Reactions.” Chemical & Engineering News, American Chemical Society, 6 Oct. 2025, cen.acs.org/safety/lab-safety/Peptide-coupling-agents-cause-severe/98/web/2020/01#:~:text=Peptide%20coupling%20agents%20help%20form%20amide%20bonds,exposed%20to%20them%20to%20develop%20an%20allergy.
“BPC-157: Experimental Peptide Creates Risk for Athletes.” NPC Hello, 25 Sept. 2025, www.usada.org/spirit-of-sport/bpc-157-peptide-prohibited/.
Understanding the Legal Risks of BPC-157 and Other Unapproved Peptides - Holt Law, djholtlaw.com/understanding-the-legal-risks-of-bpc-157-and-other-unapproved-peptides/.
by, Written. “BPC-157 Dosage Guide: How Much Should You Take for Recovery and Injury.” Swolverine, swolverine.com/blogs/blog/bpc-157-dosage-guide-how-much-should-you-take-for-recovery-and-injury-healing?srsltid=AfmBOopEQ5GQz5x5XI-Iv9faY96ORQ3snFUZ_94q2VwNaLsPaJUiD-RU.
The PrepTide: Disclaimer

