SEMAGLUTIDE
Semaglutide selectively mimics the action of the natural incretin hormone GLP-1 (Glucagon-like Peptide 1). Unlike many fat-loss peptides or thermogenic supplements that work by stimulating the nervous system, semaglutide acts on metabolic pathways and hunger-regulating centers in the brain, making it uniquely effective for long-term fat reduction and body recomposition.

SEMAGLUTIDE OVERVIEW
Category: GLP-1 Receptor Agonist, Peptide Hormone Analog
How It Works: Semaglutide selectively mimics the action of the natural incretin hormone GLP-1 (Glucagon-like Peptide 1). Unlike many fat-loss peptides or thermogenic supplements that work by stimulating the nervous system, semaglutide acts on metabolic pathways and hunger-regulating centers in the brain, making it uniquely effective for long-term fat reduction and body recomposition.
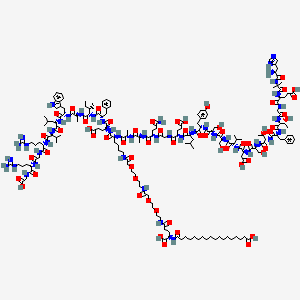
Chemical Structure: H-His-Ala-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Val-Ser-Ser-Tyr-Leu-Glu-Gly-Gln-Ala-Ala-Lys-Glu-Phe-Ile-Ala-Trp-Leu-Val-Arg-Gly-Arg-Gly-OH
Alternative Names: Ozempic, Wegovy, Rybelsus
CAS Number: 910463-68-2
WHAT IS SEMAGLUTIDE
Semaglutide is a synthetic peptide analog of the naturally occurring hormone GLP-1 (glucagon-like peptide-1), an endogenous hormone released by the gut in response to food intake. It is a highly successful and widely prescribed medication used to manage Type 2 Diabetes and obesity.
-
Mechanism: Originally designed as a treatment for Type 2 Diabetes, semaglutide mimics the effects of GLP-1 by enhancing insulin secretion, slowing gastric emptying, and reducing appetite.
- Result: These combined effects result in improved blood sugar control and, more importantly for many significant weight loss.
Brand Names
Semaglutide is currently sold under several brand names for specific indications:
-
Ozempic: Approved for Type 2 Diabetes.
-
Wegovy: FDA-approved specifically for chronic weight management.
-
Rybelsus: The first oral semaglutide formulation.
WHAT DOES SEMAGLUTIDE DO?
Unlike many fat-loss peptides or thermogenic supplements that work by stimulating the nervous system, semaglutide acts on metabolic pathways and hunger-regulating centers in the brain, making it uniquely effective for long-term fat reduction and body recomposition.
Semaglutide acts as a selective agonist of the GLP-1 receptor, mediating effects across multiple organ systems.
Multi-System Effects
Semaglutide's action provides three key, synergistic benefits:
-
Satiety Enhancement (Central Action): GLP-1R activation in the hypothalamus and brainstem reduces appetite, decreases food cravings, and enhances feelings of fullness (satiety), leading to significantly decreased caloric intake.
-
Glycemic Control (Pancreatic Action): It activates GLP-1R on pancreatic beta-cells to stimulate glucose-dependent insulin secretion and suppresses alpha-cell glucagon release. This results in lower blood sugar levels with a low risk of hypoglycemia.
-
Gastric Emptying Delay: It slows the rate at which food leaves the stomach, prolonging the feeling of fullness and reducing post-meal glucose spikes.
BENEFITS/ CLINICAL TRIALS
Semaglutide is one of the most clinically validated drugs in modern metabolic medicine, with benefits extending far beyond glucose control into body composition, performance, and long-term metabolic health.
FDA Approved Indications
-
Chronic Weight Management (Wegovy): Approved for use in adults with obesity or those who are overweight with at least one comorbidity.
-
Type 2 Diabetes (Ozempic/Rybelsus): Significantly lowers HbA1c levels and fasting plasma glucose.
-
Cardiovascular Risk Reduction: Approved to reduce the risk of MACE (major adverse cardiovascular events, including CV death, nonfatal MI, nonfatal stroke) in patients with T2D or obesity/overweight and established cardiovascular disease.
Key Clinical Trial Findings
-
Significant Weight Loss (STEP Trials): Clinical trials (STEP-1 and STEP-3) demonstrated a mean weight loss of approximately 15 percent or greater in total body weight over 68 weeks at the 2.4 mg dose (Wegovy) in non-diabetic patients. This reduction is sustained for up to 4 years and positions semaglutide well above older GLP-1 agonists.
- Cardiovascular Protection (SELECT Trial): In patients with established Cardiovascular Disease (CVD) and obesity/overweight, Semaglutide was associated with a statistically significant 20 percent reduction in MACE during a 40-month period. This benefit was observed regardless of the amount of weight lost, suggesting GH-independent effects on vascular health and inflammation.
Comprehensive Metabolic Benefits
Semaglutide's action on GLP-1 receptors provides multiple synergistic health benefits:
-
Appetite Suppression and Craving Reduction: Semaglutide suppresses hunger signals and improves satiety, leading users to report decreased hunger between meals, fewer food cravings (especially sugar and junk food), and increased control over portion sizes and emotional eating.
-
Improved Insulin Sensitivity: Even in individuals without diabetes, semaglutide improves insulin signaling and fasting glucose levels, which enhances nutrient partitioning and reduces the likelihood of storing excess calories as fat.
-
Cardiometabolic Health: Semaglutide may improve several key biomarkers, including lower LDL cholesterol and triglycerides, reduced blood pressure, and decreased CRP and systemic inflammation.
-
Investigational Benefits: It improves lipid profiles and is being investigated for efficacy in kidney disease and fatty liver disease.
SIDE EFFECTS
While semaglutide is considered safe for most users, especially when dosed properly, it does come with a list of common side effects, most of which are digestive in nature and often temporary.
Common Side Effects
Gastrointestinal (GI) side effects are the most common adverse events, reflecting the drug's mechanism of slowing gastric emptying and acting on CNS satiety centers. These effects are dose-dependent and typically diminish over time with proper dose titration.
-
Digestive (Very Common): Nausea, vomiting, constipation or diarrhea, abdominal pain, and bloating.
-
Appetite and General: Loss of appetite, mild fatigue or dizziness.
-
Note: These side effects are most common during the first 4–8 weeks of use and tend to resolve as your body adjusts. Starting at a low dose and gradually titrating up is the best way to minimize them.
Serious but Rare Risks
-
Acute Organ Risks: Acute pancreatitis and acute gallbladder disease (cholelithiasis or cholecystitis) have been reported in the drug class.
-
Thyroid Risk: Semaglutide is contraindicated in patients with a personal or family history of Medullary Thyroid Carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). This is based on rodent data showing a risk of C-cell tumors; human risk is not yet confirmed.
-
Renal Risk: Acute kidney injury has been reported, often associated with dehydration resulting from severe vomiting or diarrhea.
-
Severe Allergic Reactions: Severe allergic reactions are a possibility, as with any medication.
IS SEMAGLUTIDE SAFE?
Semaglutide is a well-studied, FDA-approved drug for specific metabolic and cardiovascular indications, supported by rigorous Phase 3 trials.
Regulatory Status and Legal Use
-
FDA Approval: Semaglutide is FDA-approved under the brand names Ozempic and Wegovy for the treatment of Type 2 Diabetes and obesity, respectively.
-
Prescription Only: In the United States, semaglutide is classified as a prescription-only medication.
-
Off-Label Use: Its use for performance enhancement or non-medical weight loss remains off-label and should only be pursued under the guidance of a licensed healthcare provider.
- Compounded Versions: Possessing or using compounded or research versions (like semaglutide acetate) without medical oversight may carry legal or health risks. The laws vary by country, so always consult local regulations.
Safety Takeaway
-
Established Safety: Safety is established when used under medical supervision, with adherence to the mandated titration schedule (slow dose increase) and strict exclusion criteria (Medullary Thyroid Carcinoma, MTC, or MEN 2).
-
Monitoring: Due to its prolonged half-life, its use requires careful monitoring for gastrointestinal (GI) side effects, particularly during the initial dose escalation.
DOSAGE
Semaglutide requires a strict, gradual titration schedule to minimize side effects, as this approach allows the body to adjust to the compound and reduces the incidence of nausea and bloating.
Administration and Maximum Doses
-
Administration: Typically administered via Subcutaneous (SubQ) injection once weekly (Ozempic, Wegovy) or as a daily oral tablet (Rybelsus).
-
Maximum Therapeutic Dose (Wegovy): The maximum dose for weight loss is 2.4 mg SubQ weekly.
-
Oral Dosing (Rybelsus): Initial dose is 3 mg daily, titrated up to a maximum of 14 mg daily. The oral formulation uses an absorption enhancer (SNAC) to enable absorption across the stomach lining.
Gradual Titration Schedule (SubQ Injection)
The titration schedule takes 16 weeks to reach the target 2.4 mg maintenance dose.
-
Starting Dose (Weeks 1–4): Most protocols begin with 0.25 mg once weekly. This low dose is critical for minimizing side effects.
-
Dose Escalation (Weeks 5+): Doses are generally increased every 4 weeks as tolerated:
-
Weeks 5–8: 0.5 mg once weekly.
-
Weeks 9–12: 1.0 mg once weekly.
-
Weeks 13+: 1.7 mg to 2.4 mg once weekly (the target maintenance dose).
-
RECONSTITUTION
Semaglutide is supplied in commercially manufactured, pre-filled pen injectors. No user reconstitution is required for the injectable forms.
Commercial Supply and Storage
-
Form: Supplied as a sterile, ready-to-use liquid solution in a multi-dose pen (Ozempic) or a single-use pre-filled pen (Wegovy).
-
Storage: Requires refrigeration as specified by the manufacturer.
Compounding Context (Hypothetical Dilutions)
While the commercial drug is pre-filled, compounded or research-grade semaglutide requires specific dilution if supplied as a powder. These examples illustrate the calculation needed to achieve a target concentration:
-
For 5 mg: Adding 2.5 mL of bacteriostatic water results in a specific concentration per mL.
-
For 15 mg: To achieve a common concentration of 1.5 mg/mL, a specific volume of bacteriostatic water is added.
-
For 30 mg: A 30 mg vial requires a custom amount of water based on the desired final concentration and individual dose. This could involve adding volumes like 7.5 mL to achieve different concentrations. Your healthcare provider will specify the exact volume.
WHERE TO BUY SEMAGLUTIDE
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for Semaglutide, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of Semaglutide outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- by, Written. “Semaglutide for Beginners: How It Works, Benefits, Side Effects, and S.” Swolverine, swolverine.com/blogs/blog/semaglutide-for-beginners-how-it-works-benefits-side-effects-and-safe-use?_pos=1&_sid=e6ddca647&_ss=r. Accessed 25 Nov. 2025.
- How Much Bacteriostatic Water to Add to 15 Mg? Tirz 15mg. ..., www.justanswer.com/medical/s0nux-bacteriostatic-water-add-15-mg-tirz-15mg.html. Accessed 25 Nov. 2025.
- “Peptide Dosage Calculator – Easily Calculate Your Peptide Doses.” Cellgenic, 15 Jan. 2025, cellgenic.com/peptide-calculator/#:~:text=Step%201:%20Set%20Your%20Dose,Check%20the%20Visual%20Dosage%20Meter.
- “Semaglutide.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/Semaglutide. Accessed 25 Nov. 2025.
-
accessdata.fda.gov. "$\text{Semaglutide}$ $\text{Subcutaneous}$ $\text{Route}$ ($\text{Ozempic}^{\text{TM}}$) $\text{Clinical}$ $\text{Pharmacology}$ $\text{Review}$."
-
Cleveland $\text{Clinic}$. "$\text{GLP}-1$ $\text{Agonists}$: $\text{What}$ $\text{They}$ $\text{Are}, \text{How}$ $\text{They}$ $\text{Work}$ $\&$ $\text{Side}$ $\text{Effects}$."
-
Mayo $\text{Clinic}$. "$\text{Semaglutide}$ ($\text{subcutaneous}$ $\text{route}$) - $\text{Side}$ $\text{effects}$ $\&$ $\text{dosage}$."
-
ACC $\text{Journal}$ $\text{Abstract}$. "$\text{Semaglutide}$ $\text{Effects}$ on $\text{Cardiovascular}$ $\text{Outcomes}$ in $\text{People}$ $\text{With}$ $\text{Overweight}$ $\text{or}$ $\text{Obesity}$ ($\text{SELECT}$ $\text{Trial}$)."
-
The $\text{Linus}$ $\text{Pauling}$ $\text{Institute}$. "$\text{Pantothenic}$ $\text{Acid}$."
-
NCBI $\text{StatPearls}$. "$\text{Semaglutide}$."
The PrepTide: Disclaimer

