MT-2
Melanotan II (MT-II) works by activating various melanocortin receptors (MC1R, MC3R, MC4R, MC5R) throughout the body, including the central nervous system. This peptide is highly non-selective, which accounts for its diverse range of effects.
MT-2 OVERVIEW
Category: Synthetic Cyclic Heptapeptide, Non-Selective Melanocortin Agonist
How It Works: Melanotan II (MT-II) works by activating various melanocortin receptors (MC1R, MC3R, MC4R, MC5R) throughout the body, including the central nervous system. This peptide is highly non-selective, which accounts for its diverse range of effects.
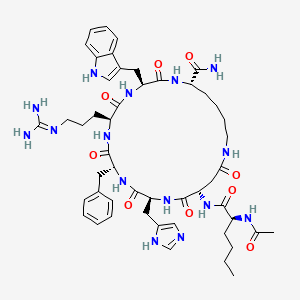
Chemical Structure: Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-NH2 (8 AAs, cyclic 2-7 lactam)
Alternative Names: TruTan, Melanotan 2, Barbie Drug
CAS Number: 121062-08-6
WHAT IS MT-2
Melanotan II (MT-II) is a synthetic, highly potent cyclic heptapeptide that acts as a non-selective agonist for the Melanocortin Receptor (MCR) family. It was developed after Melanotan I (Afamelanotide).
-
Parent Hormone: MT-II is a synthetic analog of the naturally occurring alpha-melanocyte-stimulating hormone (alpha-MSH)
-
Development Goal: It was engineered to create a compound with greater potency and stability than the native alpha-MSH hormone, primarily to stimulate melanin production for skin tanning.
WHAT DOES MT-2 DO?
Melanotan II works by activating various melanocortin receptors (MC1R, MC3R, MC4R, MC5R) throughout the body, including the central nervous system (CNS). MT-2's diverse effects are due to its non-selective binding to multiple melanocortin receptor subtypes.
Non-Selective Multi-Receptor Action
-
Pigmentation (MC1R): Activation of MC1R on melanocytes stimulates melanogenesis and the production of protective eumelanin, leading to skin darkening (tanning) without extensive UV exposure.
-
Sexual Function (MC3R, MC4R): Activation of MC4R in the CNS is responsible for its powerful effect on sexual arousal and the initiation of penile erection. This mechanism is central-acting, unlike peripheral drugs (for EX: Sildenafil).
-
Appetite and Metabolism (MC3R, MC4R): CNS activation of these receptors leads to anorexigenic (appetite suppression) effects and modulates energy homeostasis.
-
Adrenal Axis (MC2R): Unlike the highly selective MT-1, MT-2 exhibits some affinity for MC2R (the ACTH receptor), which can directly stimulate Cortisol release from the adrenal cortex.
BENEFITS/ CLINICAL TRIALS
Melanotan II (MT-II) was studied primarily for two distinct, non-cosmetic applications before research was halted. It is an unlicensed, unapproved substance globally.
-
Skin Tanning (Melanogenesis): MT-II successfully induces rapid skin darkening at lower cumulative doses than MT-1. It stimulates melanocytes to produce melanin, the pigment responsible for skin color, leading to a darker complexion without extensive UV exposure. Two subjects in a pilot study had increased pigmentation in the face, upper body, and buttocks just one week after MT-II dosing ended.
-
Sexual Function (Erectile Dysfunction or ED): MT-II activates pro-erectile pathways in the brain and nervous system, and has been studied as a potential treatment for male erectile dysfunction.
-
Small clinical trials demonstrated that MT-II was a potent initiator of penile erection in men with both psychogenic and organic ED. Subjects reported significant erectile rigidity and increased sexual desire following injection.
-
A stretching and yawning complex appeared to correlate with the onset of spontaneous penile erections, which were intermittently experienced for 1 to 5 hours after MT-II dosing.
-
-
Appetite and Weight Loss: MT-II can suppress appetite and has demonstrated anorectic effects in animal models.
Clinical Trial Details (Pilot Study)
A pilot Phase I study demonstrated MT-II's tanning activity in humans given only five low doses every other day by subcutaneous injection.
-
Dosing: The starting dose was 0.01 mg/kg. The recommended single MT-II dose for future Phase I studies was 0.025 mg/kg per day.
-
Adverse Effects: Mild nausea, not requiring antiemetic treatment, was reported at most MT-II dose levels. Grade II somnolence and fatigue were noted in one subject at the 0.03 mg/kg dose.
Regulatory Status and Research History
-
Development History: MT-II was initially developed for tanning but was quickly repurposed for ED research after its strong sexual side effects were discovered. This line of research led to the development of Bremelanotide (PT-141), a more selective MC3R/MC4R agonist.
-
FDA Status: MT-II is not FDA approved for any indication and is an unlicensed, unapproved substance globally.
SIDE EFFECTS
Melanotan II is an unlicensed and unapproved drug for human use as a tanning agent in many countries, including Australia. Health agencies strongly warn against its use because the full range of potential side effects is unknown.
MT-II carries a substantially higher risk profile than the milder MT-I due to its non-selective action on receptors throughout the body and central nervous system (CNS).
Severe Acute and Systemic Effects
-
Priapism (CRITICAL): Spontaneous, painful, and prolonged penile erections that can last for hours. Case reports detail individuals requiring emergency medical intervention to resolve priapism caused by Melanotan use.
-
Gastrointestinal Upset: Severe and frequent nausea, vomiting, stomach cramps, and facial flushing, often requiring medication to manage.
-
Cardiovascular/Renal: Reported side effects include increased blood pressure, systemic toxicity, kidney dysfunction, and potential renal infarction (tissue death).
-
Endocrine/Metabolic: Clinical use has been associated with hypercortisolism (elevated Cortisol), which can destabilize blood sugar and electrolyte balance, particularly in individuals with underlying conditions like Type 1 Diabetes.
Oncological and Dermal Risks
-
Dermal Changes: Development of new moles (eruptive nevi) and darkening or changes in the structure of existing moles (dysplastic nevi).
-
Melanoma Risk: Health authorities warn that MT-II abuse coincides with sun exposure and carries an unknown, theoretical risk of promoting or accelerating melanoma due to its inherent mitogenic action (cell-dividing effect) on melanocytes.
IS MT-2 SAFE?
Melanotan II (MT-II) is POSSIBLY UNSAFE for human use and is banned worldwide. It is sold as an illegal research chemical. Promoters of these tanning products often claim they are safe and natural, but this is false; they are far from safe.
Severe Risks and Consequences
-
Unapproved and Unassessed: Melanotan is not approved for sale or use as a tanning agent in Australia or by the FDA. These tanning agents haven’t been assessed for their quality or safety. Since they are unapproved, you have no way of knowing what's truly in them, no matter what's written on the label. They could be made with toxic, poor quality, or counterfeit ingredients.
-
Most Concerning Side Effect: The most concerning risk is the potential for serious skin cancers. MT-II has been associated with reports of increased moles and freckles and the darkening of existing nevi.
-
Systemic Toxicity: The peptide's potent and unpredictable side effects, stemming from its non-selective activation of receptors, include:
-
Kidney dysfunction and swelling of the brain.
-
Priapism (prolonged, painful penile erection) and potential complications related to Cortisol elevation.
-
Common side effects include headache, nausea, vomiting, loss of appetite, and facial redness.
-
Safety Takeaway
-
Extreme Hazard: Unsupervised use of MT-II is extremely hazardous. Health agencies in the US, UK, and Australia have issued explicit warnings against its use for cosmetic purposes.
-
Action Required: If you have recently used unapproved tanning products containing melanotan, you should stop immediately and speak to a health professional.
-
False Protection: No tan, fake or real, will protect skin against damage from sun exposure. Use of melanotan to increase skin pigmentation artificially will not protect against UV exposure the way a suitable sunscreen will.
DOSAGE
Dosing for Melanotan II is non-standardized and highly empirical, often based on maximizing tanning effects while trying to avoid severe nausea and priapism.
Administration
-
Administration: Typically administered via subcutaneous (SubQ) injection.
-
Cycle Length: Typically administered until desired pigmentation is achieved (often 1 to 2 weeks), followed by infrequent maintenance dosing.
Example Empirical Dosing Guidelines
-
Loading Dose (First Two Weeks): Inject 10 units (200 mcg) SQ daily for the first two weeks.
-
Maintenance Dose: Inject 5 units (100 mcg) two to three times per week thereafter to maintain pigmentation. Adjust accordingly to pigment changes.
-
Research/Off-Label Range: Protocols suggest a range of 0.01 mg/kg to 0.025 mg/kg daily.
-
Timing: The peptide is often taken shortly before bedtime to mitigate daytime side effects like nausea and flushing.
RECONSTITUTION
MT-II is supplied as a lyophilized (freeze-dried) powder.
-
Reconstitution Fluid: Use sterile or bacteriostatic water (BAC Water) or saline.
-
Custom Dilution (EX: 10 mg Vial): Reconstitute with 4–5 mL of BAC water for 10 mg of MT-II, depending on the concentration desired.
-
Standard Dilution (High Concentration): For a 10 mg vial, adding 1 mL of BAC water results in a highly concentrated stock solution of 10 mg/mL.
WHERE TO BUY MT-2
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for MT-2, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of MT-2 outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- Melanotan II - an Overview | Sciencedirect Topics, www.sciencedirect.com/topics/neuroscience/melanotan-ii. Accessed 24 Nov. 2025.
- “Advice on the Use of Injecting Tanning Agents, e.g. Melanotan.” DRUGS.Ie - Drug and Alcohol Information Ireland, www.drugs.ie/news/article/advice_on_the_use_on_injecting_tanning_agents_melanotan#:~:text=What%20are%20the%20side%2Deffects?%20As%20this%20is,and%20increased%20blood%20pressure%20have%20been%20reported. Accessed 23 Nov. 2025.
- Dorr RT;Lines R;Levine N;Brooks C;Xiang L;Hruby VJ;Hadley ME; “Evaluation of Melanotan-II, a Superpotent Cyclic Melanotropic Peptide in a Pilot Phase-I Clinical Study.” Life Sciences, U.S. National Library of Medicine, pubmed.ncbi.nlm.nih.gov/8637402/#:~:text=Two%20subjects%20had%20increased%20pigmentation,NCI%20NIH%20HHS/United%20States. Accessed 23 Nov. 2025.
- Peptide Worksheet, www.majesticaestheticswellness.com/wp-content/uploads/2023/05/Peptide-Worksheet-.pdf. Accessed 24 Nov. 2025.
- “Melanotan-II.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/melanotan-II. Accessed 23 Nov. 2025.
-
Actas $\text{Dermo-Sifiliográficas}$. "$\text{Melanotan}$ $\text{II}$ $\text{and}$ $\text{Its}$ $\text{Clinical}$ $\text{Implications}$."
-
ResearchGate (ID 12289786). "$\text{Melanocortin}$ $\text{receptor}$ $\text{agonists}, \text{penile}$ $\text{erection}, \text{and}$ $\text{sexual}$ $\text{motivation}$: $\text{Human}$ $\text{studies}$ $\text{Melanotan}$ $\text{II}$."
-
PubMed Central (PMC6388891). "$\text{Melanotan-induced}$ $\text{priapism}$: a $\text{hard-earned}$ $\text{tan}$."
-
PubMed (ID 9679884). "$\text{Synthetic}$ $\text{melanotropic}$ $\text{peptide}$ $\text{initiates}$ $\text{erections}$ $\text{in}$ $\text{men}$ with $\text{psychogenic}$ $\text{erectile}$ $\text{dysfunction}$."
-
Amazing $\text{Meds}$. "$\text{GHK-Cu}$ $\text{Side}$ $\text{Effects}$: $\text{Risks}, \text{Safety}, \text{and}$ $\text{Precautions}$."
-
ResearchGate (ID 364242115). "$\text{Safety}$ $\text{and}$ $\text{efficacy}$ of a $\text{GLP}-1$ $\text{and}$ $\text{glucagon}$ $\text{receptor}$ $\text{dual}$ $\text{agonist}$ $\text{mazdutide}$ ($\text{IBI}362$) $\text{9 mg}$ and $\text{10 mg}$..."
The PrepTide: Disclaimer

