SLU-PP-332
SLU-PP-332 is a synthetic research compound and peptide mimetic of exercise currently in early-stage research for its potential in treating metabolic disorders, such as obesity and Type 2 Diabetes. Its potent action is rooted in its ability to bind to and activate ERR nuclear receptors (primarily ERR alpha), which triggers a genetic program in skeletal muscle identical to that induced by aerobic endurance exercise.

SLU-PP-332 OVERVIEW
Category: Estrogen Related Receptor Agonist, Exercise Mimetic
How It Works: SLU-PP-332 is a synthetic research compound and peptide mimetic of exercise currently in early-stage research for its potential in treating metabolic disorders, such as obesity and Type 2 Diabetes. Its potent action is rooted in its ability to bind to and activate ERR nuclear receptors (primarily ERR alpha), which triggers a genetic program in skeletal muscle identical to that induced by aerobic endurance exercise.
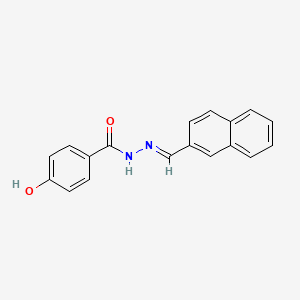
Chemical Structure: C18 H14 N2 O2
Alternative Names: Exersize Mimetic Drug, ERR Pan Agonist
CAS Number: 303-760-60-3
WHAT IS SLU-PP-332
SLU-PP-332 is a synthetic small molecule compound, not a peptide, currently in early-stage research for its potential in treating metabolic disorders, such as obesity and Type 2 Diabetes. It is not approved for human consumption and is sold for laboratory use only.
-
Classification: SLU-PP-332 is one of the most promising compounds in the emerging field of exercise mimetics, which aims to provide the cellular metabolic benefits of physical activity through pharmacology.
-
Target: It was developed to act as an agonist for the Estrogen Related Receptor (ERR) family of orphan nuclear receptors.
-
Cellular Action: The interesting thing about SLU-PP-332 is that it doesn't work like your typical fat burner. Instead of cranking up your heart rate or suppressing your appetite, this compound focuses on how your body actually handles fat at the cellular level. Think of it as teaching your body to be more efficient with the energy it already has.
WHAT DOES SLU-PP-332 DO?
SLU-PP-332 functions by mimicking the molecular signals produced during high-intensity endurance training, primarily within skeletal muscle. It acts as an agonist for the Estrogen Related Receptor (ERR) family, enhancing both mitochondrial function and energy metabolism.
Key Cellular Actions
-
ERR Activation: The compound binds to ERR alpha and ERR gamma, which are transcription factors that regulate the expression of key metabolic genes. The compound binds to the ligand binding domain of ERR alpha, stabilizing the receptor’s active conformation and strengthening its interaction with coactivator proteins, especially PGC-1 alpha (peroxisome proliferator activated receptor gamma coactivator 1 alpha).
-
Mitochondrial Biogenesis: Activation of these receptors stimulates the production of new mitochondria and increases the expression of OXPHOS (Oxidative Phosphorylation) enzymes and Cytochrome C, enhancing cellular respiration and energy efficiency.
-
Fatty Acid Oxidation: SLU-PP-332 shifts whole-body metabolism toward favoring fat as a primary fuel source. It increases the proportion of oxidative (slow twitch) muscle fibers (Type IIa), which are fatigue-resistant and excel at burning fat.
-
Energy Expenditure: The activation of this aerobic exercise genetic program leads to increased energy expenditure (thermogenesis) even at rest, causing fat loss without changes in food intake.
Through its impact on fat oxidation, glucose utilization, and thermogenesis, SLU-PP-332 shows promise as a treatment avenue for metabolic disorders.
BENEFITS/ CLINICAL TRIALS
Research on SLU-PP-332 is focused on translating the benefits of endurance exercise into a therapeutic treatment for patients unable to exercise due to physical limitations, age, or chronic disease. This synthetic compound works behind the scenes by adjusting how your body processes energy, offering a non-stimulant approach to metabolic health.
Key Benefits Observed in Preclinical Models
-
Endurance Enhancement: Mice administered SLU-PP-332 showed a dramatic increase in running distance (up to 45 percent) and running time (up to 70 percent) without changing their physical activity levels. This boost comes from teaching the body to be more efficient with the energy it already has.
-
Obesity Treatment and Fat Loss: Obese mouse models treated with SLU-PP-332 lost approximately 12 percent of their total body weight, gained 10 times less fat than untreated mice, and showed improvements in glucose metabolism and insulin sensitivity while maintaining a stable appetite.
-
This encourages fat use instead of storage, supporting leaner physique goals.
-
-
Metabolic Health: Improves metabolic flexibility, making it easier for the body to switch between carbs and fats, which is important for blood sugar control and long term metabolic balance. Early findings also suggest a protective effect against fat buildup in the liver (reducing fatty liver risk).
-
Tissue Protection: Research suggests potential for treating metabolic diseases like Type 2 Diabetes and Heart Failure (HFpEF) due to the compound's ability to enhance mitochondrial function in the heart and kidneys.
-
Muscle Phenotype Shift: Converts muscle fibers to a more oxidative (endurance-like) phenotype, reducing fat mass while maintaining lean muscle.
Lifestyle Applications
-
Non-Stimulant Alternative: Perfect for those who do not tolerate caffeine or harsh thermogenics but still want fat loss support.
-
Energy Efficiency: Instead of quick spikes and crashes, SLU-PP-332 could support steadier, longer lasting energy output.
Clinical Trial Findings and Future Status
-
Formal Clinical Trials: SLU-PP-332 has not yet entered human clinical trials and remains strictly a preclinical research compound.
-
Future Focus: The next step in research is optimizing the compound structure for oral bioavailability.
SIDE EFFECTS
Since SLU-PP-332 has not been tested in humans, the true safety profile is unknown. Preclinical data, however, indicates specific safety concerns related to its pan-ERR agonism (activation of multiple Estrogen Related Receptors).
Preclinical Toxicity Concerns (Animal Models)
-
Cardiac Hypertrophy: The ERR gamma receptor is expressed in the heart. ERR activation was linked to cardiac hypertrophy (enlargement of heart cells) in some mouse models, raising caution for chronic use.
-
Hepatotoxicity: Very high doses (greater than or equal to 100 mg/kg) caused transient elevations in liver enzymes (ALT/AST) in mice, though this usually resolved.
-
Nutrient Exhaustion: Chronic dosing depleted muscle glycogen reserves, suggesting a risk of nutrient exhaustion under continuous, uncontrolled stimulation.
Observed Human Effects (Theoretic)
-
Digestive Changes: Because it shifts how fats are processed, some users may notice mild gastrointestinal (GI) discomfort like bloating or changes in digestion.
-
Lipid Profile Shifts: Any compound that influences fat metabolism could theoretically change cholesterol or triglyceride levels, though more research is needed to confirm this.
-
Metabolic Adaptation: As with most compounds, long term use may cause the body to adapt, reducing effectiveness if not cycled intelligently.
Observed Safety in Mice
-
Acute Safety: Acute toxicity studies showed no obvious toxicity and no change in spontaneous food intake or appetite.
-
Unknowns with Extended Use: Most of the data so far is preclinical, meaning long term human safety has not been established.
IS SLU-PP-332 SAFE?
SLU-PP-332 is an unapproved, high-risk preclinical research compound. Since it is still in the research stage, its safety profile is not as well defined as established compounds. All current usage is experimental, and safety in humans is not established.
Critical Safety Takeaway
-
Classification: SLU-PP-332 is strictly classified as a research chemical.
-
High Risk: Due to its potent effect on mitochondrial proliferation and the associated preclinical risks of cardiac hypertrophy (enlargement of heart cells), it cannot be recommended for any non-research application.
-
Usage Limitation: Its use is limited to studies investigating its ERR mechanism of action.
DOSAGE
Dosing protocols are based on maximizing therapeutic windows in animal models, not human patients. All human-equivalent dosing is empirical and not backed by clinical trials.
Preclinical (Animal) Context
-
Administration: Typically administered via Intraperitoneal (IP) injection or in animal feed during preclinical studies.
-
Dosing Range: Highly effective doses in mice were typically 50 mg/kg administered twice daily (b.i.d.).
-
Frequency: Twice-daily (b.i.d.) administration was necessary to maintain consistent exposure and efficacy.
Empirical Administration (Human Context)
-
Administration Route: Typically given via subcutaneous injection, similar to many other peptides.
-
Dosage Range (Reported): Early reports suggest anywhere from 1–5 mg per week, though this is not clinically validated.
-
Frequency:
-
Some protocols split the dose into multiple injections throughout the week to keep levels stable.
-
Others use larger, less frequent injections.
-
-
Tolerance Testing: Those exploring it in research often start at the low end of the range to monitor response.
-
Stacking: May pair well with other fat-loss peptides like AOD-9604 or Tesamorelin, though research is very limited.
Timing Considerations
-
Before Training: Some experiment with dosing prior to endurance workouts to maximize fat utilization.
-
Cycling: Because the body can adapt to metabolic shifts, SLU-PP is often discussed in the context of cycling (EX: 4–8 weeks on, followed by time off).
RECONSTITUTION
SLU-PP-332 is a small molecule supplied as a powder. Due to its unique chemical properties, it requires specialized solvents and storage conditions that differ from standard peptides.
Reconstitution and Solvents
-
Reconstitution Fluid: It is highly soluble in organic solvents. It is typically dissolved in a DMSO/Tween/PBS solution for research injection, or PEG400 for animal oral administration. It is generally not reconstituted with pure Bacteriostatic Water.
-
Example Dilution: A common reconstitution is to add 3.0 mL bacteriostatic water to the vial, which yields an approximate 1.67 mg/mL concentration (if a 5 mg vial is assumed).
Storage and Stability (CRITICAL)
-
Powder Storage: The powder should be stored long term at -20°C or -80°C.
-
Solution Stability: Reconstituted solutions have limited stability and must be stored frozen to maintain efficacy.
WHERE TO BUY SLU-PP-332
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for SLU-PP-332, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of SLU-PP-332 outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- What Is SLU-PP-332? How Does It Work?, www.peptidesciences.com/peptide-research/what-is-slu-pp-332-how-does-it-work. Accessed 25 Nov. 2025.
- by, Written. “SLU-PP Peptide: Benefits, Dosage, and Metabolic Effects Explained.” Swolverine, swolverine.com/blogs/blog/slu-pp-peptide-benefits-dosage-and-metabolic-effects-explained. Accessed 25 Nov. 2025.
- “SLU-PP-332 (5mg Vial) Dosage Protocol.” Peptidedosages.Com, 14 Nov. 2025, peptidedosages.com/single-peptide-dosages/slu-pp-332-5mg-vial-dosage-protocol/.
- “SLU-PP-332.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine,
pubchem.ncbi.nlm.nih.gov/compound/5338394. Accessed 25 Nov. 2025.
- PubMed Central (PMC11584170). "Synthetic $\text{ERR}\alpha/\beta/\gamma$ $\text{Agonist}$ $\text{Induces}$ $\text{an}$ $\text{ERR}\alpha\text{-Dependent}$ $\text{Acute}$ $\text{Aerobic}$ $\text{Exercise}$ $\text{Response}$ $\text{and}$ $\text{Enhances}$ $\text{Exercise}$ $\text{Capacity}$."
- UF News. "$\text{Exercise-mimicking}$ $\text{drug}$ $\text{sheds}$ $\text{weight}, \text{boosts}$ $\text{muscle}$ $\text{activity}$ in $\text{mice}$."
- ResearchGate (ID 394764077). "$\text{Mazdutide}$ $\text{reduces}$ $\text{body}$ $\text{weight}$ $\text{in}$ $\text{adults}$ $\text{with}$ $\text{overweight}$ $\text{or}$ $\text{obesity}$: $\text{A}$ $\text{high-dose}$ $\text{Phase }1$ $\text{trial}$."
- J Ren Endocrinol. "$\text{New}$ $\text{hopes}$ $\text{on}$ '$\text{SLU-PP-332}$' $\text{as}$ $\text{an}$ $\text{effective}$ $\text{agent}$ $\text{for}$ $\text{weight}$ $\text{loss}$ $\text{with}$ $\text{indirect}$ $\text{kidney}$ $\text{protection}$ $\text{efficacy}$.
- MDPI (ID 1422-0067/20/10/2456). "$\text{Mitochondrial-Derived}$ $\text{Peptide}$ $\text{MOTS-c}$ $\text{Increases}$ $\text{Adipose}$ $\text{Thermogenic}$ $\text{Activation}$ $\text{to}$ $\text{Promote}$ $\text{Cold}$ $\text{Adaptation}$."
The PrepTide: Disclaimer

