IGF-1LR3
IGF-1 LR3 is a potent peptide recognized in the realm of fitness and wellness for its ability to accelerate growth and recovery. It is highly valued for its performance-enhancing and regenerative properties.
IGF-1LR3 OVERVIEW
Category: Recombinant Polypeptide Hormone, Anabolic Growth Factor Analog
How It Works: IGF-1 LR3 is a potent peptide recognized in the realm of fitness and wellness for its ability to accelerate growth and recovery. It is highly valued for its performance-enhancing and regenerative properties.
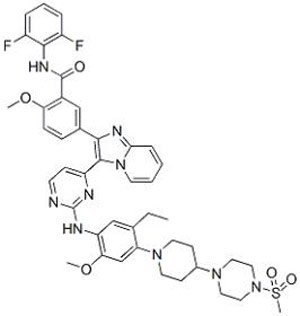
Chemical Structure: C500 H625 N111 O115 S9
Alternative Names: Long R3-IGF-1
CAS Number: 143045-27-6
WHAT IS IGF-1LR3
IGF-1 LR3 (Insulin-like Growth Factor-1 Long Arginine 3) is a synthetic, non-endogenous analog of the naturally occurring human hormone IGF-1. IGF-1 is the primary mediator of the effects of Growth Hormone (GH) and is responsible for most of the body's growth, anabolic (tissue-building), and recovery processes.
Structure and Enhanced Stability
IGF-1 LR3 is a synthetic peptide hormone designed to mimic and enhance the effects of natural IGF-1, a critical growth factor involved in muscle development, recovery, and cellular repair.
-
Structure: It is structurally modified to contain 83 amino acids (natural IGF-1 has 70).
-
Stability: This modification gives IGF-1 LR3 a significantly longer half-life, approximately 20 to 30 hours, compared to natural IGF-1, which typically lasts only 12–15 minutes in circulation.
-
Action: This long half-life allows it to drive muscle hyperplasia (new cell creation) and hypertrophy (cell size increase) over a sustained period by binding to the IGF-1 Receptor (IGF-1R) with high affinity.
WHAT DOES IGF-1LR3 DO?
IGF-1 LR3 functions as a potent, long-acting agonist of the IGF-1 Receptor (IGF-1R), activating key intracellular signaling pathways, primarily the PI3K Akt and MAPK pathways.
Enhanced Anabolism
Hormone-like Growth Factor 1 (IGF-1) is naturally synthesized in the liver in response to growth hormone (GH) stimulation. IGF-1 LR3, however, can be administered exogenously to bypass this indirect process and produce faster results.
-
Bypasses Regulation: Its design limits its affinity for IGF-binding proteins (IGFBPs), which normally restrict IGF-1's activity. This modification allows IGF-1 LR3 to remain free-floating in the bloodstream and exert more direct anabolic effects.
-
Signaling and Synthesis: The prolonged, unbound presence of IGF-1 LR3 in the bloodstream maximizes the anabolic signaling effect on muscle tissue, activating intracellular signaling pathways that promote protein synthesis and muscle fiber hypertrophy (increase in cell size).
Muscle Growth and Nutrient Partitioning
-
Muscle Hyperplasia: IGF-1 LR3 is strongly linked to the activation and proliferation of satellite cells (muscle stem cells). This process, known as hyperplasia, leads to the creation of new muscle fibers and increased muscle cell numbers, contributing to long-term growth potential.
-
Nutrient Partitioning: Due to its structural and functional homology with Insulin, IGF-1 LR3 enhances glucose and amino acid transport into muscle cells while simultaneously promoting lipolysis (fat breakdown) in adipose tissue. This enhanced nutrient uptake favors muscle gain over fat storage.
BENEFITS/ CLINICAL TRIALS
The research interest in IGF-1 LR3 is primarily focused on its enhanced anabolic (tissue-building) and lipolytic (fat-burning) potency compared to the native hormone.
Key Anabolic and Systemic Benefits
IGF-1 LR3's anabolic effects make it a powerful tool for enhanced growth and recovery.
-
Muscle Growth and Strength Gains: Promotes significant lean muscle mass accrual and increased strength, particularly when combined with resistance training. It stimulates the proliferation and differentiation of satellite cells, which are essential for muscle repair and development, driving both muscle hyperplasia and hypertrophy.
-
Enhanced Recovery: Promotes faster repair of damaged tissues post-training, speeds up tissue repair, enhances collagen synthesis, and reduces recovery time from injury or intense exercise.
-
Improved Fat Metabolism (Fat Loss): Supports fat oxidation and improves overall metabolism, leading to reduced body fat mass and better nutrient partitioning.
-
Bone Density: Mouse model studies have also linked IGF-1 signaling to improvements in bone density and skeletal strength, suggesting systemic health benefits beyond just muscular development.
-
Cognitive and Cardiovascular Support: Low endogenous IGF-1 levels are linked to increased risk of cardiovascular and cognitive decline; IGF-1 LR3 is hypothesized to mitigate these risks by supporting neuronal survival and improved cardiac output.
Research and Regulatory Status
-
Approved Use (Native IGF-1): The native recombinant human IGF-1 (Mecasermin) is FDA-approved for growth failure in children with severe IGF-1 deficiency.
-
IGF-1 LR3 Trials: IGF-1 LR3 itself is not FDA-approved for any human use. It has been used in small, non-clinical research contexts.
-
Risk Note: For example, Infusion studies in fetal sheep models found that high doses of IGF-1 LR3 were found to induced severe hypoglycemia and hypoxemia. This highlights the potency and risk of misuse of the compound.
SIDE EFFECTS
The side effects of IGF-1 LR3 are directly related to its potent anabolic and metabolic effects. They are generally more pronounced and longer-lasting than those seen with short-acting peptides due to its prolonged half-life.
Metabolic and Endocrine Risks
-
Hypoglycemia (CRITICAL RISK): The most serious acute risk is low blood sugar. IGF-1 LR3 transports glucose out of the bloodstream into muscle cells, potentially causing dangerous drops in blood sugar, especially when injected in a fasted state. Symptoms include dizziness, weakness, confusion, and irritability. To reduce the risk, users should monitor blood sugar, especially during the first few weeks of use, and consume adequate carbohydrates with each dose.
-
Insulin Resistance: Chronic, high-dose use can downregulate insulin receptors, potentially leading to long-term insulin resistance and elevated fasting glucose.
Musculoskeletal and Structural Concerns
-
Acromegaly-like Symptoms: Abuse or high-dose use can lead to abnormal, irreversible soft tissue and bone growth. This includes enlarged hands, feet, jaw, and internal organ enlargement (for example: heart, liver).
-
Water Retention: Temporary fluid retention and bloating can lead to mild water retention, causing a bloated appearance, and in some cases, elevated blood pressure. These effects are generally short-term.
-
Joint Pain/Stiffness: Some users report joint pain or stiffness (arthralgia), particularly when using higher doses. This may result from accelerated muscle growth, placing stress on joint structures.
-
Local Reactions: Injection site pain or discomfort.
IS IGF-1LR3 SAFE?
Despite the purported effects on athletic performance, serious health risks are associated with a healthy individual abusing IGF-1 LR3.
Severe Health Risks
-
Acromegaly-like Effects: Similar to Growth Hormone (GH) abuse, IGF-1 LR3 abuse can lead to acromegaly, a long-term condition in which body tissues get larger over time. Non-reversible side effects to the heart, joints, and liver may occur due to abnormal growth.
-
Cancer Risk: IGF-1 LR3 is one of the riskiest compounds discussed in regenerative research due to its potent mitogenic (cell division) properties. While beneficial for normal growth, it poses a theoretical risk of accelerating the growth of existing or dormant cancers (for example, prostate, breast, colon) by inhibiting apoptosis (programmed cell death) and driving cell replication.
Regulatory Status and Safety Takeaway
-
FDA Status: IGF-1 LR3 is not approved by the FDA for human therapeutic use and is highly restricted.
-
Safety Takeaway: Due to its significant risks, particularly the risk of accelerating cancer growth, its use is strictly discouraged outside of controlled laboratory research.
DOSAGE
Dosing for IGF-1 LR3 is based on anecdotal data and preclinical research, aiming for low-dose, infrequent administration due to the compound's potency and long half-life. Precision and individualization are essential.
Administration and Frequency
-
Administration: Typically administered via subcutaneous (SubQ) injection.
-
Administration Frequency: Due to its long half-life of 20 to 30 hours, it is typically administered once daily or 5 days per week (for EXAMPLE: excluding rest days).
Dosing Guidelines and Timing
Most protocols recommend a dosage between 20 to 100 micrograms (mcg) per day, depending on factors such as body weight, fitness goals, and previous peptide use.
-
General Daily Dosage: Protocols suggest extremely low doses, ranging from 20 mcg to 80 mcg per day.
-
Beginner Dose: For beginners, it is advisable to start at the lower end (20–40 mcg/day) to assess tolerance and minimize side effects.
-
Advanced Use: Some bodybuilders and performance athletes may gradually increase to higher dosages during short cycles, but long-term use without medical oversight is discouraged due to potential systemic effects.
-
Cycle Length: Short cycles of 4 to 6 weeks are common to avoid receptor desensitization and long-term side effects.
Critical Timing and Hypoglycemia Risk
-
Injection Timing: Many users choose to inject post-workout or before bedtime to align with natural growth hormone (GH) surges. Post-workout timing may enhance nutrient partitioning and recovery, while bedtime dosing may promote overnight repair and regeneration.
-
Hypoglycemia Warning: Injection should NEVER be done while fasted. Adequate carbohydrate intake (25 to 50 g) immediately post-injection is essential to prevent hypoglycemia due to the compound's potent insulin-mimicking effect.
RECONSTITUTION
IGF-1 LR3 is supplied as a lyophilized (freeze-dried) powder. Proper reconstitution technique is crucial to maintain the hormone's potency.
Reconstitution Fluid and Dilution
-
Reconstitution Fluid: Use sterile or bacteriostatic water (BAC Water). IGF-1 LR3 can also be reconstituted with dilute acetic acid for long-term stability, but BAC water is common for immediate use.
-
Standard Dilution (EX: 1 mg Vial): For a 1 mg vial, adding 1 mL of BAC water results in a standard concentration of 1 mg/mL (1,000 mcg/mL). This allows for precise drawing of the microgram doses recommended for administration.
Mixing Technique
The goal is to dissolve the delicate peptide structure without damaging it.
-
Prepare Diluent: Pull an amount of air into the mixing syringe equal to the amount of diluent you plan to add (for EXAMPLE: 1 mL) to create negative pressure. Insert the needle into the diluent vial and inject the air. Then, draw the desired amount of liquid into the syringe.
-
Inject Diluent: Slowly inject the diluent into the peptide powder vial. Aim the stream of liquid at the side of the glass wall, not directly onto the powdered peptide, to prevent damage to the delicate protein structure.
-
Mix Gently: Once all the liquid is in the vial, remove the syringe. DO NOT SHAKE vigorously or vortex. Instead, gently swirl or roll the vial between your hands for a few minutes (around 4 minutes) until all the powder is completely dissolved and the solution is clear.
-
Aliquot for Storage (Optional): To ensure stability and avoid repeated thawing, you can apportion the reconstituted solution into smaller, single-use or weekly-use aliquots in separate sterile vials.
Post-Reconstruction Storage
-
Storage: Once reconstituted, IGF-1 LR3 must be stored in the refrigerator (2°C to 8°C)
-
Stability: The reconstituted solution is generally stable for 1 to 3 months, depending on the formulation and diluent used.
WHERE TO BUY IGF-1LR3
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for IGF-1LR3, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of IGF-1LR3 outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- Https://Www.Sciencedirect.Com/Science/Article/Abs/Pii/S1047847720300046?Via=ihub, www.med.upenn.edu/pmi/events/https-www-sciencedirect-com-science-article-abs-pii-s1047847720300046-via-3dihub. Accessed 17 Nov. 2025.
- “IGF-1 and the World Anti-Doping Agency Prohibited List: USADA.” NPC Hello, 9 Apr. 2014, www.usada.org/spirit-of-sport/igf-1-and-the-world-anti-doping-agency-prohibited-list/#:~:text=Despite%20the%20purported%20effects%20on,joints%2C%20and%20liver%20may%20occur.
- by, Written. “Unlocking the Power of IGF-1 LR3: The Ultimate Guide to Growth and Rec.” Swolverine, swolverine.com/blogs/blog/unlocking-the-power-of-igf-1-lr3-the-ultimate-guide-to-growth-and-recovery?_pos=1&_sid=210a34ab7&_ss=r. Accessed 17 Nov. 2025.
- Https://Www.Sciencedirect.Com/Science/Article/Abs/Pii/S1047847720300046?Via=ihub, www.med.upenn.edu/pmi/events/https-www-sciencedirect-com-science-article-abs-pii-s1047847720300046-via-3dihub. Accessed 21 Nov. 2025.
- “Insulin-like Growth Factor I (57-70).” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/insulin-like-growth-factor-I-_57-70. Accessed 21 Nov. 2025.
- “Recombinant Human IGF-I/IGF-1 DES (1-3) Protein.” Novus Biologicals, www.novusbio.com/products/igf-i-igf-1-recombinant-protein_nbp2-34947#:~:text=Reconstitute%20in%20sterile%20distilled%20water,0.1%2D1.0mg/mL. Accessed 21 Nov. 2025.
-
The Journal of Clinical Endocrinology & Metabolism. "Prolonged stimulation of growth hormone ($\text{GH}$) and insulin-like growth factor I secretion by $\text{CJC}-1295$, a long-acting analog of $\text{GH}$-releasing hormone, in healthy adults."
-
Pediatric Research. "$\text{IGF}-1 \text{ LR}3$ for Fetal Growth Restriction."
-
PubMed Central (PMC). "Optimizing $\text{IGF}-1$ for skeletal muscle therapeutics."
-
ChemicalBook. "Mechanism and Side Effects of $\text{IGF}-1 \text{ LR}3$."
-
Swolverine. "Side Effects of $\text{IGF}-1$ Peptides: What to Watch Out For."
-
USADA. "$\text{IGF}-1$ and the World Anti-Doping Agency Prohibited List."
-
Diet vs Disease. "$\text{IGF}-1$: Miracle Hormone or Health Hazard?"
The PrepTide: Disclaimer

