KPV
KPV acts as a heavy hitter in regulating immune response and calming inflammation. It does this by inhibiting the activation of the master inflammatory transcription factor within cells, thereby suppressing the production of pro-inflammatory cytokines.

KPV OVERVIEW
Category: Tripeptide a-MSH Fragment, Selective Anti-Inflammatory
How It Works: KPV acts as a heavy hitter in regulating immune response and calming inflammation. It does this by inhibiting the activation of the master inflammatory transcription factor within cells, thereby suppressing the production of pro-inflammatory cytokines.
Chemical Structure: Lys-Pro-Val
Alternative Names: a-MSH(11-13)
CAS Number: 1458-86-2
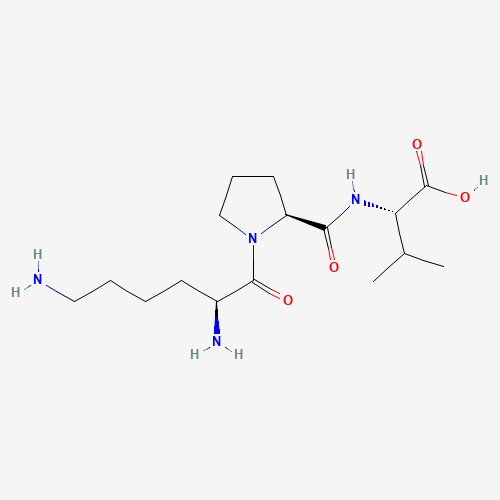
WHAT IS KPV
KPV (Lysine-Proline-Valine) is a tripeptide (three amino acids) derived from the C-terminal sequence of alpha-Melanocyte Stimulating Hormone (alpha-MSH).
-
Parent Hormone: Alpha-MSH is a larger, naturally occurring hormone known for its wide-ranging protective, anti-inflammatory, and immune-modulating effects.
-
Targeted Action: Despite its small size, KPV packs an outsized punch in regulating immune responses and calming inflammation. KPV retains the core anti-inflammatory power of the parent hormone but lacks the pigmentation-inducing activity of full-length alpha-MSH.
-
Mechanism: Unlike many anti-inflammatory drugs that carry significant side effects, KPV works more subtly—guiding the body’s own systems back into balance.
WHAT DOES KPV DO?
KPV (Lysine-Proline-Valine) is a tripeptide composed of just three amino acids: Lysine (K), Proline (P), and Valine (V). It is derived from the larger peptide hormone alpha-Melanocyte Stimulating Hormone (alpha-MSH), a key regulator in immune and inflammatory responses.
Structure and Classification
-
Structure: Small, stable, and bioactive. Its simplicity allows it to be delivered in multiple forms, including oral, topical, or injectable research preparations.
-
Classification: KPV belongs to the family of melanocortin-derived peptides, with activity tied to the melanocortin receptor system (especially MC1R and MC3R).
-
Purpose: While alpha-MSH has wide-ranging effects, the KPV fragment retains strong anti-inflammatory properties without the hormonal baggage (like pigmentation-inducing activity) of its parent molecule.
Central Mechanism: NF-kB Inhibition
KPV is a potent immune and inflammation regulator, capable of acting both systemically and locally at tissue barriers.
-
NF-kB Inhibition (Master Regulator): The central mechanism is the dose-dependent inhibition of the activation of NF-kB. NF-kB is a critical transcription factor that drives the expression of numerous pro-inflammatory genes. By blocking its activation and nuclear import, KPV shortens the duration of the inflammatory response.
-
Cytokine Suppression: This NF-kB blockade leads to a significant downregulation of major pro-inflammatory cytokines, such as TNF-alpha, IL-1 beta, and IL-6, which fuel chronic inflammation.
-
Targeted Cellular Uptake: KPV can be taken up by cells via the PepT1 transporter, which is normally found in the small intestine but is highly upregulated in the colon during states of inflammation (such as IBD). This suggests a natural, targeted delivery mechanism to inflamed gut tissue.
-
Antimicrobial and Barrier Protection: KPV exhibits direct antimicrobial activity against common pathogens (for example, Staphylococcus aureus, Candida albicans) and supports the integrity of the intestinal and cutaneous epithelial barriers.
BENEFITS/ CLINICAL TRIALS
KPV has attracted attention because of its wide-ranging effects on inflammation control, tissue repair, and immune balance. Unlike blunt anti-inflammatories, it works as a regulator—restoring equilibrium without fully suppressing the immune system.
KPV stands out because it can be applied orally, topically, or systemically, making it one of the most versatile research peptides in the inflammation and repair category.
Key Benefits Observed in Research Models
-
Supports Barrier Integrity: Supports intestinal barrier integrity, reducing the risk of "leaky gut."
-
Calms Bowel Responses: Calms inflammatory bowel responses, showing potential in research models of colitis and Crohn’s disease (Inflammatory Bowel Disease or IBD). In mouse models of colitis, oral KPV reduced inflammatory infiltrates, suppressed inflammatory cytokine expression (TNF-alpha), and led to earlier recovery.
-
Reduces Systemic Inflammation: May reduce systemic inflammation linked to poor gut health by preventing bacterial translocation and toxin leakage.
-
Accelerates Healing: Accelerates wound closure by reducing local inflammation and supporting tissue remodeling.
-
Skin Conditions: Studied for use in eczema, psoriasis, and dermatitis, where overactive inflammation drives flare-ups.
-
Local Action: When applied topically, it may help reduce redness, swelling, and irritation while promoting healing.
-
Antimicrobial Action: Demonstrated bactericidal and candidacidal effects in research, indicating potential for use in wounds with coexisting infection.
-
Cytokine Suppression: Suppresses inflammatory cytokines (TNF-alpha, IL-1 beta, IL-6) across multiple tissues.
-
Immune Modulation: Modulates the immune response without suppressing the immune system entirely, making it useful in models of chronic inflammatory and autoimmune conditions.
-
Recovery & Performance: By dampening chronic inflammation, KPV may improve recovery speed after training or injury. It reduces oxidative stress load, helping cells maintain energy balance during repair.
Clinical Trial Findings
-
Formal Clinical Trials: There are no finalized, large-scale randomized controlled trials in humans establishing the safety or efficacy of KPV for treating specific diseases (IBD, arthritis, etc.).
-
Research Focus: Current translational research focuses on developing optimal delivery methods (nanoparticles, liposomes, transdermal enhancers) to overcome its poor passive bioavailability. Oral delivery of KPV encapsulated in hyaluronic acid nanoparticles showed enhanced therapeutic efficacy in mouse colitis models.
SIDE EFFECTS
KPV is noted for its favorable acute toxicity profile, even at high doses in preclinical studies. No major systemic toxicity or hormonal side effects have been reported in existing preclinical literature.
Side Effects (Generally Mild and Transient)
-
Local/Topical: Transient skin irritation (mild redness/itching) may occur at topical application sites.
-
Gastrointestinal: Possible mild GI upset (bloating, discomfort) may occur with oral use at higher experimental doses.
-
Systemic/Anecdotal: Fatigue or headache has been mentioned anecdotally, though not consistently reported in formal studies.
Key Safety Advantage
-
Selectivity: Its lack of pigmentation activity is a major advantage over the parent alpha-MSH molecule, allowing it to be used for anti-inflammatory purposes without the side effect of skin darkening.
IS KPV SAFE?
KPV is an investigational tripeptide and is not FDA-approved for human therapeutic use.
Safety Profile
KPV has shown a very favorable safety profile in both animal models and limited human experimental use. Because it's a small, naturally occurring tripeptide fragment of alpha-MSH, it is generally well tolerated and does not appear to cause the systemic complications often associated with stronger immunosuppressing drugs.
-
Tolerability: KPV demonstrates excellent acute tolerability in research models, lacking the pigmentation activity of its parent molecule and major systemic toxicity.
-
Delivery Challenge: Its effectiveness relies heavily on advanced delivery technology (for example, nanoparticles, etc.) that is currently only proven in laboratory settings, intended to overcome its poor passive bioavailability.
Safety Takeaway
While KPV is derived from a natural host defense hormone, it cannot be recommended for routine clinical use due to the lack of independent, rigorous human clinical safety and efficacy data.
DOSAGE
Because KPV is still a research-only peptide, there are no standardized clinical dosing guidelines. What exists comes from preclinical studies and experimental protocols using oral, injectable, or topical delivery. Dosing protocols are non-standardized and highly dependent on the delivery route.
Administration Routes and Empirical Dosing
-
Administration: Available in research as Topical Cream/Gel, Oral Capsules (or nanoparticle formulations), or Subcutaneous Injection.
-
Topical Dosing: Concentrations typically range from 0.01 percent to 1 percent for wound or skin applications.
-
Oral Dosing (Empirical): Protocols suggest a range of 1 mg to 5 mg daily for gut health, often split into two doses.
-
Injectable Dosing (Empirical): Typically ranges from 0.5 mg to 2 mg per administration for systemic effects.
Preclinical (Animal) Dosing
-
Preclinical Studies: Studies often use 1–5 mg/kg bodyweight in animal models.
-
Findings: In mice with chemically induced colitis, KPV injections significantly reduced inflammation and tissue damage.
-
Note: Human-equivalent doses would be much smaller, but data remain exploratory.
RECONSTITUTION
KPV is typically supplied as a lyophilized (freeze-dried) powder for injectable or compounding purposes.
Reconstitution Fluid and Dilution
-
Reconstitution Fluid: Use sterile or bacteriostatic water (BAC Water).
-
Draw Volume: Draw the desired volume of BAC water (for EX: 1 mL or 3.0 mL) with a sterile syringe.
-
Standard Dilution (EX: 5 mg Vial): For a 5 mg vial, adding 1 mL of BAC water results in a highly concentrated stock solution of 5 mg/mL (5,000 mcg/mL).
Mixing Technique
The mixing technique is crucial to avoid damaging the peptide.
-
Injection: Inject the BAC Water slowly down the vial wall; avoid foaming.
-
Mixing: DO NOT SHAKE vigorously. Gently swirl or roll the vial until the powder is dissolved into a clear solution.
Post-Reconstitution Storage
-
Storage: Label the vial with the date of reconstitution and refrigerate the solution immediately at 2°C to 8°C (36°F to 46°F). The solution should be protected from light.
-
Stability: Once reconstituted, the solution is generally stable for 14 to 28 days.
WHERE TO BUY KPV
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for KPV, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of KPV outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- “KPV (10mg Vial) Dosage Protocol.” Peptidedosages.Com, 20 Nov. 2025, peptidedosages.com/single-peptide-dosages/kpv-10mg-vial-dosage-protocol/.
- by, Written. “KPV Peptide: Anti-Inflammatory Benefits, Mechanism, and Research Guide.” Swolverine, swolverine.com/blogs/blog/kpv-peptide-mechanism-benefits-and-research-applications?_pos=1&_sid=88547ac5f&_ss=r. Accessed 22 Nov. 2025.
- “KPV Peptide: The Anti-Inflammatory Molecule You’ve Probably Never Heard Of.” Longevity, vocal.media/longevity/kpv-peptide-the-anti-inflammatory-molecule-you-ve-probably-never-heard-of. Accessed 22 Nov. 2025.
-
The American Journal of Pathology (PMC2431115). "$\text{PepT}1\text{-Mediated}$ $\text{Tripeptide}$ $\text{KPV}$ $\text{Uptake}$ $\text{Reduces}$ $\text{Intestinal}$ $\text{Inflammation}$."
-
Frontiers in Pharmacology (PMC5498804). "Orally $\text{Targeted}$ $\text{Delivery}$ of $\text{Tripeptide}$ $\text{KPV}$ via $\text{Hyaluronic}$ $\text{Acid-Functionalized}$ $\text{Nanoparticles}$ $\text{Efficiently}$ $\text{Alleviates}$ $\text{Ulcerative}$ $\text{Colitis}$."
-
PubMed (ID 22837805). "Inhibition of cellular and systemic inflammation cues in human bronchial epithelial cells by melanocortin-related peptides: mechanism of $\text{KPV}$ action and a role for $\text{MC}3\text{R}$ $\text{agonists}$."
-
PubMed (ID 18092346). "$\text{Melanocortin-derived}$ tripeptide $\text{KPV}$ has $\text{anti-inflammatory}$ potential in murine models of $\text{inflammatory}$ $\text{bowel}$ $\text{disease}$."
-
Innerbody. "$\text{KPV}$ $\text{Peptide}$ $\text{Benefits}, \text{Safety}$ & $\text{Buying}$ $\text{Advice}$."
-
Dr.Oracle. "What are the safety and efficacy considerations for using $\text{KPV}$ ($\text{KPV}$ $\text{peptide}$) for $\text{medical}$ $\text{applications}$?"
The PrepTide: Disclaimer

