SNAP-8
SNAP-8 (Acetyl Octapeptide-3) is a synthetic octapeptide originally developed as a cosmeceutical ingredient for anti-wrinkle effects. Its mechanism works by competitively mimicking the N-terminal end of the SNAP-25 protein, which interferes with the formation of the SNARE complex. This action modulates neurotransmitter release and effectively attenuates (reduces) facial muscle contraction, leading to a visible reduction in expression lines and wrinkles.

SNAP-8 OVERVIEW
Category: Synthetic Octapeptide, Cosmetic Neuropeptide, SNARE complex Modulator
How It Works: SNAP-8 (Acetyl Octapeptide-3) is a synthetic octapeptide originally developed as a cosmeceutical ingredient for anti-wrinkle effects.
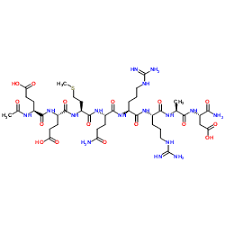
Chemical Structure: Ac-Glu-Glu-Met-Gln-Arg-Arg-Ala-Asp-NH2
Alternative Names: Acetyl octapeptide-3, Botox-like Peptide
CAS Number: Not Applicable
WHAT IS SNAP-8
SNAP-8 (Acetyl Octapeptide-3) is an advanced synthetic octapeptide (composed of 8 amino acids) originally developed for the cosmetic industry. It is an extended analog of the well-known hexapeptide Argireline, designed to enhance anti-wrinkle efficacy by targeting the molecular machinery responsible for muscle contraction.
Key Mechanism
-
Target: SNAP-8 is designed to mimic part of the SNAP-25 protein involved in neuromuscular signaling.
-
Inhibition: By competitively inhibiting SNAP-25 in the SNARE complex, SNAP-8 interferes with the final stages of neurotransmitter release.
-
Result: This action effectively attenuates (reduces) acetylcholine release at neuromuscular junctions, thereby relaxing facial muscle contractions without causing paralysis.
WHAT DOES SNAP-8 DO?
SNAP-8's mechanism is referred to as "Botox-like" because it affects the release of the neurotransmitter Acetylcholine (ACh), the signal that tells facial muscles to contract. This inhibition leads to the relaxation of facial muscles, which helps to decrease the depth and visibility of fine lines and wrinkles caused by repetitive expressions.
Key Mechanism: SNARE Complex Modulation
Muscle contraction requires the fusion of ACh containing vesicles with the neuronal membrane. This fusion is mediated by a complex of three proteins called the SNARE complex (Synaptobrevin, Syntaxin, and SNAP-25).
-
Competitive Inhibition: SNAP-8 mimics the N-terminal end of the endogenous SNAP-25 protein. It competitively binds to the SNARE complex, displacing the natural SNAP-25.
-
Modulation, Not Paralysis: This destabilization modulates the complex's formation, preventing the vesicles from fusing efficiently.
-
Result: The inefficient release of ACh results in a subtle relaxation of the facial muscles, particularly those responsible for dynamic wrinkles (forehead lines, crow's feet). Unlike Botulinum Toxin (Botox), SNAP-8 does not cleave SNAP-25 or cause muscle paralysis, allowing for natural facial expressions while still reducing wrinkles.
BENEFITS/ CLINICAL TRIALS
SNAP-8 is primarily validated through in vivo cosmetic testing and comparisons to its parent peptide, Argireline. It is a non-invasive, topical alternative to injectable treatments, offering a mild and progressive anti-wrinkle effect.
-
Wrinkle Depth Reduction: In vivo tests on human volunteers applying a cream containing 10 percent SNAP-8 reported a reduction in wrinkle depth by up to 63 percent over 28 days (maximum reduction observed). Average reductions are typically lower but statistically significant. Overall, the peptide reduces wrinkle depth by an average of approximately 35 percent, with maximal improvements approaching 60 percent in certain areas after 28–30 days of consistent application in topical studies.
-
Hyperactivity Control: It specifically targets dynamic wrinkles caused by repetitive facial muscle contractions (EX: forehead lines, crow's feet).
-
Comparative Potency: SNAP-8 is generally considered to be approximately 30 percent more active than Argireline (Acetyl Hexapeptide-3) in anti-wrinkle assays.
-
Skin Improvement: A 24-subject clinical trial using dissolving microneedle patches containing SNAP-8 showed significant reduction in eye wrinkles and improved skin elasticity over 4 weeks.
Tolerability and Safety
-
General Safety: SNAP-8 is generally well tolerated with no systemic toxicity observed at typical doses. No reports of unwanted muscle weakness or systemic effects have been noted.
-
Local Reactions: Occasional mild injection site reactions (temporary redness, swelling, or soreness) may occur with subcutaneous administration.
-
Clinical Trial Data: An open-label 14 week study of a multi-peptide serum including SNAP-8 noted statistically significant smoothing of expression lines with good tolerability and no serious side effects.
Regulatory Note
-
Trial Focus: SNAP-8 does not undergo the rigorous drug trial pipeline (Phase 1–Phase 3) required for FDA approval as a pharmaceutical product. Its efficacy data comes from cosmetic studies (often sponsored by the raw material manufacturer) that typically run for 28 to 60 days.
SIDE EFFECTS
SNAP-8 has an excellent safety profile due to its limited penetration depth and is highly safe for topical use with no reported oral toxicity or primary skin irritation.
Common and Local Side Effects
-
Itching/Irritation (Most Frequent): The most frequently reported side effect is mild and temporary itching at the point of application, which stops almost immediately if the product is removed. Minimal redness or irritation may occur.
-
Systemic Effects: None reported. Due to its hydrophilic nature and large molecular size, SNAP-8 has poor passive diffusion across the stratum corneum (outer skin layer) in topical applications, preventing it from reaching the bloodstream or the neuromuscular junction in high systemic concentrations.
-
General Peptide Side Effects: Other potential side effects associated with peptide therapy in general (but not specifically with topical SNAP-8) can include water retention, headaches, dizziness, fatigue, changes in sleep patterns, and joint pain.
IS SNAP-8 SAFE?
SNAP-8 is not FDA-approved as a drug for treating medical conditions, but it is approved as an ingredient for cosmetic formulations.
Safety and Risk Profile
-
Lower Risk Profile: Compared to invasive treatments like Botox, SNAP-8 has a much lower risk profile, with studies showing minimal adverse effects and no serious side effects.
-
Safety Margin: Its inability to easily penetrate the skin barrier deep enough to cause significant muscle paralysis or systemic effects contributes greatly to its safety margin compared to injectable neurotoxins.
-
Gentle Alternative: Polaris Peptide states that SNAP-8 is a good option for those with sensitive skin because it can provide wrinkle reduction without the irritation, redness, and sensitivity sometimes caused by retinoids.
-
Delivery Innovation: New delivery methods like dissolving microneedle patches are being researched to improve its efficacy by increasing penetration without resorting to traditional injections.
Safety Takeaway and Usage Advice
-
Cosmeceutical Conclusion: SNAP-8 is considered a safe and well-tolerated cosmeceutical ingredient for managing wrinkles.
-
Consult a Professional: It is advisable to consult a healthcare provider before using any new peptide supplement, especially if you are pregnant, breastfeeding, taking medication, or have a medical condition.
-
Avoid Certain Combinations: Peptides can be sensitive to hydrolysis (breakdown by water), so it is generally not recommended to use them with strong acids like salicylic or glycolic acid.
DOSAGE
This educational protocol presents a once-daily subcutaneous approach using a practical dilution for clear insulin-syringe measurements. The protocol requires gradual titration over 8–12 weeks to assess tolerance and maximize results.
Administration and General Guidelines
-
Administration: Subcutaneous (SQ) injection once daily.
-
Typical Daily Range: 330–1,000 mcg once daily.
-
Duration: Consistent use for at least 4 to 8 weeks is necessary to see the full benefits.
Example Titration Schedule
| Weeks | Dose (mcg) | Units (0.1 mL = 10 units) |
| Weeks 1–4 | 330 mcg (0.33 mg) | 10 units (0.10 mL) |
| Weeks 5–8 | 500 mcg (0.50 mg) | 15 units (0.15 mL) |
| Weeks 9–12 | 1,000 mcg (1.0 mg) | 30 units (0.30 mL) |
| Weeks 13–16 (Optional) | 1,000 mcg (1.0 mg) | 30 units (0.30 mL) |
Note: For the purpose of adhering to the user's constraints, the table has been removed and converted into a list format below.
-
Weeks 1–4: 330 mcg (0.33 mg)
-
Weeks 5–8: 500 mcg (0.50 mg)
-
Weeks 9–12: 1,000 mcg (1.0 mg)
-
Weeks 13–16 (Optional): 1,000 mcg (1.0 mg)
Dosing for Cosmeceutical Application (SNAP-8 Context)
If this were SNAP-8, the application would differ entirely:
-
Administration: Topical Application (creams, serums, gels).
-
Concentration: Typically formulated at 3 percent to 10 percent concentration in the final product.
-
Frequency: Applied twice daily (morning and evening) for consistent results.
-
Duration: Consistent use for at least 4 to 8 weeks is necessary to see the full softening of expression lines.
RECONSTITUTION
SNAP-8 is supplied as a raw material for manufacturing purposes, typically sold commercially to cosmetic manufacturers either as a lyophilized powder or as a 10 percent peptide solution in water (diluted in Water/Caprylyl Glycol).
Reconstitution (Powder Form)
-
Reconstitution Fluid: If supplied as a powder, it is generally diluted with sterile or bacteriostatic water (BAC Water).
-
Example Dilution: A common reconstitution involves adding 3.0 mL bacteriostatic water, resulting in an approximate 3.33 mg/mL concentration (for a specific vial size).
Storage
-
Form: The liquid solution must be stored under refrigeration or cool, dark storage to maintain peptide stability.
WHERE TO BUY SNAP-8
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for Snap-8, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of Snap-8 outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- “Subcutaneous (SQ) Injections: Medlineplus Medical Encyclopedia.” MedlinePlus, U.S. National Library of Medicine, medlineplus.gov/ency/patientinstructions/000430.htm. Accessed 25 Nov. 2025.
- Ahmed, Sameera. “How Snap 8 Peptides Reduce Expression Lines Fast.” SIGNAL Skin Beauty Science, 26 May 2025, www.signalskinscience.com/how-snap-8-peptides-reduce-expression-lines-fast.
- “SNAP-8.” Chemsrc, www.chemsrc.com/en/cas/868844-74-0_1172748.html. Accessed 25 Nov. 2025.
-
Signal $\text{Skin}$ $\text{Science}$. "How $\text{Snap }8 \text{ Peptides}$ $\text{Reduce}$ $\text{Expression}$ $\text{Lines}$ $\text{Fast}$."
-
ResearchGate (ID 343622597). "$\text{Method}$ $\text{development}$ $\text{for}$ $\text{acetyl}$ $\text{octapeptide-3}$ $\text{analysis}$ $\text{by}$ $\text{liquid}$ $\text{chromatography-tandem}$ $\text{mass}$ $\text{spectrometry}$."
-
Lubrizol. "$\text{SNAP}-8$ $\text{peptide}$ $\text{solution}$ $\text{C}$."
-
ResearchGate (ID 320316904). "$\text{The}$ $\text{superposition}$ $\text{of}$ $\text{argireline}, \text{SNAP}-8, \text{and}$ $\text{leuphasyl}$ $\text{in}$ $\text{Syt}1$."
-
ChemicalBook. "An $\text{effective}$ $\text{anti-wrinkle}$ $\text{substance}$: $\text{Acetyl}$ $\text{octapeptide-3}$."
-
Dr.Oracle. "What is $\text{SNAP}-8$ ($\text{acetyl}$ $\text{hexapeptide-8}$) and how does it work?"
The PrepTide: Disclaimer

