RETATRUTIDE
An experimental medication developed by Eli Lilly that is being studied as a next generation treatment for obesity, overwight symptoms and many other related metabolic conditions such as type 2 diabetes.

RETATRUTIDE OVERVIEW
Category: GLP-1, Triple Hormone Receptor Agonist
How It Works: Activates three incretin/metabolic hormone receptors simultaneously to amplify satiety, slow down digestion, and increase natural energy expenditure from fat burning.
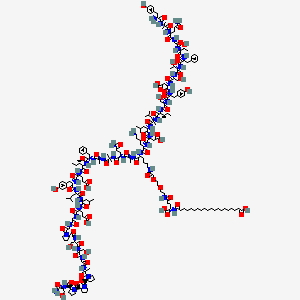
Chemical Structure: Synthetic 39 amino acid triple agonist, Proprietary Complex peptidyl structure with C20 fatty acid
Alternative Names: LY3437943
CAS Number: Not applicable
WHAT IS RETATRUTIDE
Retatrutide, also known as LY-3437943, is an experimental medication developed by Eli Lilly that is being studied as a next-generation treatment for obesity, overweight, and related metabolic conditions such as type 2 diabetes.
Retatrutide is a triple agonist. It targets GLP-1, GIP, and glucagon receptors simultaneously. This unique mechanism has shown unbelievable results in early clinical trials. Participants lost over 24% of their body weight in less than a year at the highest studied doses.
Retatrutide is a long-acting peptide analog engineered for high potency and once-weekly administration.
WHAT DOES RETATRUTIDE DO?
Retatrutide is the first molecule studied to activate all three key receptors governing metabolic health: GLP-1 (Glucagon-Like Peptide-1), GIP (Glucose-Dependent Insulinotropic Polypeptide), and GCGR (Glucagon Receptor).
-
Satiety and Appetite Suppression (GLP-1/GIP): Activation of these receptors in the brainstem and hypothalamus amplifies the feeling of fullness and significantly reduces overall food intake and cravings.
-
Gastric Regulation (GLP-1): Slows gastric emptying, which contributes to a sustained feeling of satiety and blunts post-meal glucose spikes.
-
Energy Expenditure and Fat Metabolism (GCGR): The unique addition of Glucagon receptor agonism is hypothesized to increase energy expenditure and promote lipolysis (the breakdown of stored fat). The Glucagon receptor's typical hyperglycemic effect is balanced by the robust insulin-promoting action of GLP-1 and GIP.
-
Glycemic Control (GLP-1/GIP): Promotes glucose-dependent insulin secretion and suppresses inappropriate glucagon release, leading to improved HbA1c levels.
BENEFITS/ CLINICAL TRIALS
Clinical trials have produced striking results. In Phase 2 studies, participants taking higher doses of retatrutide lost an average of up to 24% of their body weight over 48 weeks. Alongside weight reduction, the drug has been shown to improve metabolic health markers, including blood sugar control, hemoglobin A1c, blood pressure, and insulin sensitivity.
Retatrutide has demonstrated clinical efficacy exceeding currently approved single and dual agonists, with results approaching those seen in bariatric surgery.
Key Benefits Observed in Phase 2 Trials:
-
Extreme Weight Loss:
-
Participants with obesity (without T2D) taking the highest dose (12 mg) lost an average of 24.2% of their initial body weight over 48 weeks.
-
-
Body Composition Improvement:
-
DEXA scan substudies showed a significant and favorable reduction in total body fat mass (up to 26.1% reduction), while maintaining a healthy ratio of fat-to-lean mass loss.
-
-
Metabolic Control (T2D):
-
HbA1c reductions up to 2.0% compared to placebo. A high percentage of participants (up to 82%) achieved HbA1c levels below 6.5%.
-
-
Liver Health (MASLD/NAFLD):
-
Over 85% of participants with fatty liver disease achieved normalization of liver fat content (below 5%) at the highest doses, suggesting a strong therapeutic potential for metabolic dysfunction-associated steatotic liver disease.
-
-
Cardiometabolic Markers: Significant improvements in waist circumference, systolic blood pressure, and lipid profiles.
SIDE EFFECTS
The safety profile of Retatrutide is consistent with the class of incretin mimetics, characterized primarily by gastrointestinal (GI) side effects that are generally dose-dependent.
-
Gastrointestinal (Most Common): Nausea, Vomiting, Diarrhea, Constipation, and abdominal discomfort. These symptoms are most pronounced during the initial titration phase when the dose is being increased.
-
Cardiovascular: Transient, dose-dependent increases in heart rate have been observed, which is a known effect of GLP-1 agonism, requiring careful monitoring.
-
Serious Adverse Events: Pancreatitis and gallbladder issues (cholelithiasis) are rare but monitored concerns associated with the broader incretin class.
IS RETATRUTIDE SAFE?
Retatrutide is currently an investigational drug candidate in large-scale Phase 3 clinical trials. It is not approved by the FDA or any global regulatory body for prescription or therapeutic use.
Short- to mid-term clinical data suggest the drug is generally well-tolerated, with adverse events being manageable and rarely leading to discontinuation when administered with proper titration. However, the full extent of long-term safety (beyond one year), including effects on bone density and chronic cardiovascular outcomes, remains under investigation pending the completion of Phase 3 trials.
DOSAGE
In non-diabetic participants with obesity, dosing was directly proportional to weight reduction. Higher weekly doses led to progressively greater fat loss:
→ 1–4 mg weekly produced modest weight loss, often in the 8–12% range.
→ 8 mg weekly resulted in significantly more reduction, approaching the magnitude seen with Trizepatide at its highest doses.
→ 12 mg weekly drove the most dramatic outcomes, with participants achieving up to 24% body weight reduction at 48 weeks — a figure exceeding results from current FDA-approved GLP-1 or GIP agonists.
|
Week Range |
Weekly Dose |
Notes on Titration & Tolerability |
|
Weeks 1–4 |
2–4 mg |
Introductory dose to minimize nausea and GI upset; allows adaptation. |
|
Weeks 5–8 |
4–8 mg |
Intermediate step; stronger weight loss effects begin to appear; GI symptoms may peak here. |
|
Weeks 9–12 |
8 mg |
Considered a “therapeutic dose” with significant weight loss outcomes and manageable side effects. |
|
Weeks 13+ |
12 mg |
Highest studied dose; associated with the greatest weight loss (~24% at 48 weeks) but also higher GI burden. |
RECONSTITUTION
A 10 mg vial of lyophilized retatrutide powder, the standard recommendation is to use 1.0 mL of bacteriostatic water. This creates a final concentration of 10 mg/mL, which is easy to dose accurately with an insulin syringe.
- 20mg use 2.0mL
- 30mg use 3.0mL
- etc.
WHERE TO BUY RETATRUTIDE
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for Retatrutide, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of Retatrutide outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- “Swolverine Top Fitness Blog: Bodybuilding, Nutrition, Supplements.” Swolverine, swolverine.com/blogs/blog/tagged/peptides. Accessed 21 Nov. 2025.
- “Retatrutide.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/Retatrutide. Accessed 21 Nov. 2025.
-
The New England Journal of Medicine. "Triple-Hormone-Receptor Agonist Retatrutide for Obesity - $\text{A Phase }2 \text{ Trial}$."
-
The Lancet Diabetes & Endocrinology. "Effects of Retatrutide on body composition in people with type $2 \text{ diabetes}$."
-
VCU Health. "Retatrutide wiped out fat in liver of obese patients."
-
MDPI. "Retatrutide—A Game Changer in Obesity Pharmacotherapy."
-
American Diabetes Association. "Highlights Novel Agent Retatrutide which Results in Substantial Weight Reduction..."
-
Expert opinion on investigational drugs. "Is Retatrutide ($\text{LY}3437943$), a $\text{GLP-1}, \text{GIP}, \text{and glucagon receptor agonist a step forward}$ in the treatment of diabetes and obesity?"
The PrepTide: Disclaimer

