PINEALON
This naturally occurring compound consists of a proprietary sequence of amino acids specifically designed to restore and optimize pineal gland function. The compound directly interacts with DNA sequences to modulate gene expression. The overall action reduces oxidative stress in brain tissue, supporting the function of the pineal gland.

PINEALON OVERVIEW
Category: Synthetic Tripeptide, Neuroprotective Agent, Cytogenetic Peptide
How It Works: This naturally occurring compound consists of a proprietary sequence of amino acids specifically designed to restore and optimize pineal gland function. The compound directly interacts with DNA sequences to modulate gene expression. The overall action reduces oxidative stress in brain tissue, supporting the function of the pineal gland.
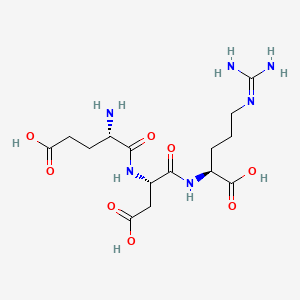
Chemical Structure: Glu-Asp-Arg
Alternative Names: EDR Peptide
CAS Number: 409740-19-4
WHAT IS PINEALON
Pinealon (EDR Peptide) is a synthetic tripeptide (composed of three amino acids: Glutamic Acid Aspartic Acid Arginine) originally synthesized based on the regulatory peptides found in the pineal gland.
Pinealon is a bioactive peptide derived from pineal gland tissue that belongs to the class of cytogenetic peptides.
Origin and Purpose
-
Natural Analog: This naturally occurring compound consists of a proprietary sequence of amino acids specifically designed to restore and optimize pineal gland function.
-
Targeted Approach: Originally developed from pineal gland extracts, Pinealon represents a targeted approach to addressing age related decline in circadian rhythm regulation and cognitive function.
-
Mechanism Class: It is primarily researched for its ability to regulate gene expression and maintain cellular homeostasis in glandular and neuronal tissues, functioning as a circadian rhythm regulator and neuroprotective agent.
WHAT DOES PINEALON DO?
Pinealon's activity is concentrated in the central nervous system (CNS) and is hypothesized to work directly at the level of DNA and core neurochemical pathways, offering neuroprotection and recovery support. This peptide has gained considerable attention in neurology and age-related medicine.
Mechanism of Action
Pinealon exhibits unique selectivity for pineal gland tissue and maintains bioactivity across various physiological conditions, making it distinctive among neurologically active peptides.
-
Gene Expression Modulation (Cytogenetic Action): Research suggests Pinealon interacts with DNA sequences to modulate the expression of genes involved in neuronal function, repair, and differentiation (EX: Nestin, beta Tubulin III). This action is theorized to help restore optimal gene function in aged or stressed neurons.
-
Antioxidant Defense: It promotes the synthesis and activity of endogenous antioxidant enzymes (in cerebellar neurons) and directly reduces the accumulation of Reactive Oxygen Species (ROS) and subsequent apoptosis (cell death) induced by oxidative stress.
-
Neuroprotection Mechanism: Pinealon has been shown to counteract damage caused by toxins (EX: Homocysteine), which are linked to Alzheimer's and vascular pathology. This protective effect may be linked to regulating the ERK1/2 (extracellular signal-regulated kinase) pathway, vital for neuronal survival.
Key Benefits
Due to its action on gene expression and antioxidant systems, Pinealon has demonstrated benefits in:
-
Cognitive Enhancement and recovery support.
-
Sleep Optimization (circadian rhythm regulation).
-
Neurotransmitter Balance: Studies indicate that Pinealon can stimulate Serotonin expression in cells of the brain cortex and may enhance neuroplasticity (synaptic connections).
BENEFITS/ CLINICAL TRIALS
Pinealon has been widely studied by its originators for its application in aging and neurological trauma, often as an oral capsule form used as an adjunct to conventional therapy. Pinealon exerts its therapeutic effects through multiple interconnected molecular pathways centered on pineal gland function restoration.
Mechanism of Action and Systemic Regulation
Pinealon's activity is hypothesized to work directly at the level of DNA and core neurochemical pathways, offering neuroprotection and recovery support.
-
Gene Expression Modulation (Cytogenetic Action): Research suggests Pinealon interacts with DNA sequences to modulate the expression of genes involved in neuronal function, repair, and differentiation.
-
Neuroprotection and Antioxidant Defense: It promotes the synthesis and activity of endogenous antioxidant enzymes and directly reduces the accumulation of Reactive Oxygen Species (ROS) and subsequent apoptosis (cell death) induced by oxidative stress. It protects against neurotoxic substances and age related damage while promoting neuronal survival pathways and synaptic maintenance.
-
Neurotransmitter System Optimization: It supports acetylcholine production for cognitive function, enhances serotonin synthesis and receptor sensitivity, optimizes dopamine pathway activity, and balances GABA ergic inhibitory neurotransmission.
Key Therapeutic Benefits
-
Cognitive Enhancement: Studies confirmed that Pinealon protects neuronal cells from hypoxia (low oxygen) and chemical neurotoxicity (Homocysteine), resulting in improved spatial orientation and learning ability. It improves short term and long term memory, attention, and cognitive function in elderly subjects with age related decline, and improves recovery of memory and perceptional motor responses following craniocerebral trauma.
-
Circadian Rhythm Regulation: It helps synchronize the natural sleep wake cycle by restoring melatonin production and regulation, enhancing circadian rhythm entrainment mechanisms, and optimizing light dark cycle responsiveness.
-
Neuroendocrine Regulation: It supports hypothalamic pituitary adrenal (HPA) axis optimization, growth hormone release regulation and enhancement, cortisol response modulation, and overall hormonal homeostasis restoration.
-
Cellular Resilience: Reduces the spontaneous cell death rate and increases the functional activity of the main cellular elements in brain tissue.
Clinical Trial Findings and Safety
Clinical trials supporting Pinealon are typically small human clinical trials stemming from non-Western research groups.
-
Limited Scope Findings: Small human clinical trials have supported the use of oral Pinealon capsules (for EXAMPLE: 0.2 mg twice daily for 20 to 30 days) as an adjunct treatment, showing reported improvements in speech, memory, and headache frequency.
-
Acute Toxicity: No toxicity has been reported in research (up to 10 mg tested), with no adverse changes noted in major organs.
-
Adverse Events: Adverse events are minimal, primarily consisting of vivid dreams and mild injection reactions. Tolerability has been excellent in extended studies.
SIDE EFFECTS
Pinealon is consistently reported in research literature as having an excellent safety profile with extremely low toxicity. Acute and chronic toxicity studies in animal models have not shown any detrimental systemic effects, even at high doses.
Common and Transient Side Effects
Any reported side effects are generally mild and transient.
-
Neurological/Sleep: Headache, dizziness, transient fatigue, mild insomnia (especially if taken too late in the day), or occasional vivid dreams/altered sleep patterns (due to increased alertness).
-
Gastrointestinal: Mild nausea or gastrointestinal discomfort.
-
General: Mild anxiety or transient mood changes during the initial adjustment period.
Local and Rare Reactions
-
Injection Site Reactions: Pain, redness, itching, swelling, or minor bruising at the injection site.
-
Allergic Reactions: As with any substance, allergic reactions (EX: hives, swelling, difficulty breathing) are possible in rare cases.
Regulatory Status and Critical Cautions
-
FDA Status: Pinealon is not an FDA-approved drug, and its long-term safety profile is not fully established through large-scale clinical trials.
-
Seizure Risk: It should be used with caution in individuals with epilepsy or seizure disorders, as central nervous system-active agents may lower the seizure threshold in rare cases.
-
Pregnancy/Breastfeeding: Safety in pregnancy and breastfeeding has not been established.
IS PINEALON SAFE?
Pinealon is currently an investigational peptide and is not FDA-approved for human therapeutic use.
Need for Medical Supervision
The lack of FDA approval and need for additional clinical research necessitate careful consideration and medical supervision for any therapeutic application.
-
Patients interested in Pinealon therapy should engage in comprehensive discussions with qualified healthcare providers to evaluate potential benefits against risks and explore evidence-based treatment alternatives.
-
Until comprehensive large-scale trials are completed, its use must remain under appropriate medical oversight with careful monitoring of treatment response and potential adverse effects.
Safety Takeaway and Future Research
-
Safety Profile: While research suggests a favorable safety profile due to its tripeptide structure and minimal systemic burden, its cytogenetic properties (gene modulation) and long-term central nervous system (CNS) effects have not been verified by independent Phase 3 Randomized Controlled Trials (RCTs).
-
Future Outlook: Future research will be critical in establishing standardized dosing protocols, confirming long-term safety, and defining optimal clinical applications of Pinealon in human medicine.
-
Conclusion: Use must remain strictly confined to research protocols.
DOSAGE
Dosing protocols for Pinealon vary based on the delivery route (oral vs. injection) and therapeutic target. Recommended administration often utilizes short, intermittent cycles to maintain optimal regulatory function.
Administration and Dosing Protocols (Research)
-
Administration Routes: Pinealon is available in research as Oral Capsules or Subcutaneous (SubQ) injection.
-
A Common Regimen (Injectable): A common regimen is 100 mcg to 300 mcg subcutaneously once daily for 10–20 days.
-
Intense SubQ Protocol: Protocols often use 5 mg per day SubQ for 10 to 20 consecutive days.
-
Oral Dosing (Research): Dosing is typically 0.1 mg to 0.2 mg once or twice daily.
-
Cycle Length: Cycles are repeated every few months as needed. Some protocols recommend combination therapy with other neuropeptides for synergistic effects.
RECONSTITUTION
Pinealon is supplied as a lyophilized (freeze-dried) powder for injection or compounding into oral capsules.
Reconstitution Fluid and Dilution
-
Reconstitution Fluid: Use sterile or bacteriostatic water (BAC Water).
-
Mixing Technique: Inject the BAC Water slowly down the side of the vial. DO NOT SHAKE vigorously. Gently swirl the vial until the powder is completely dissolved into a clear solution.
-
Example Dilution Protocols (Based on a 10 mg Vial):
-
For 1 mg per 1 mL: Add 10 mL of BAC water. This means every 10 units on a U-100 syringe equals 1 mg (1,000 mcg) of Pinealon.
-
For 2 mg per 1 mL: Add 5 mL of BAC water. This means every 10 units on a U-100 syringe equals 0.5 mg (500 mcg) of Pinealon.
-
For 4 mg per 1 mL: Add 2.5 mL of BAC water. This means every 10 units on a U-100 syringe equals 0.25 mg (250 mcg) of Pinealon.
-
Standard Stock: Add 1 mL of BAC water. This results in a highly concentrated stock solution of 10 mg/mL.
-
Post-Reconstitution Storage
-
Storage: Once reconstituted, the solution must be stored in the refrigerator (2°C to 8°C).
-
Stability: The solution is generally stable for 14 to 28 days under proper refrigeration.
WHERE TO BUY PINEALON
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for Pinealon, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of Pinealon outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- “Pinealon Peptide: Brain & Memory Support.” Peptide | Brain & Memory Support, www.paragonsportsmedicine.com/peptides/pinealon#:~:text=Pinealon%20is%20a%20bioactive%20peptide,mechanisms%20with%20the%20pineal%20gland. Accessed 24 Nov. 2025.
- “Pinealon.” Recess Rx, recessrx.com/pinealon#:~:text=COMMON%20SIDE%20EFFECTS,breastfeeding%20has%20not%20been%20established. Accessed 24 Nov. 2025.
- Pinealon: A Powerful Peptide for Cellular Rejuvenation, enhancedwellnessny.com/wp-content/uploads/2025/02/Pinealon-A-Powerful-Peptide-for-Cellular-Rejuvenation-.pdf. Accessed 24 Nov. 2025.
- “Glu-Asp-Arg.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/Glu-Asp-Arg. Accessed 24 Nov. 2025.
-
ResearchGate (ID 348139472). "$\text{EDR}$ $\text{Peptide}$: $\text{Possible}$ $\text{Mechanism}$ of $\text{Gene}$ $\text{Expression}$ and $\text{Protein}$ $\text{Synthesis}$ $\text{Regulation}$ $\text{Involved}$ $\text{in}$ $\text{the}$ $\text{Pathogenesis}$ of $\text{Alzheimer}'\text{s}$ $\text{Disease}$."
-
Enhanced Wellness $\text{NY}$. "$\text{Pinealon}$: $\text{A}$ $\text{Powerful}$ $\text{Peptide}$ $\text{for}$ $\text{Cellular}$ $\text{Rejuvenation}$."
-
PubMed Central (PMC3342713). "$\text{Pinealon}$ protects the $\text{rat}$ $\text{offspring}$ $\text{from}$ $\text{prenatal}$ $\text{hyperhomocysteinemia}$."
-
MDPI (ID 1424-8247/14/6/515). "$\text{Neuroprotective}$ $\text{Effects}$ of $\text{Tripeptides}—\text{Epigenetic}$ $\text{Regulators}$ in $\text{Mouse}$ $\text{Model}$ of $\text{Alzheimer}'\text{s}$ $\text{Disease}$."
-
Paragon Sports Medicine. "$\text{Pinealon}$ $\text{Peptide}$ $\text{Brain}$ $\&$ $\text{Memory}$ $\text{Support}$."
-
Age $\text{Management}$ $\text{Medicine}$ $\text{Group}$. "$\text{Epitalon}$ $\text{Pinealon}$ $\text{Dosing}$."
The PrepTide: Disclaimer

