PT-141
PT-141 (Bremelanotide) selectively activates Melanocortin Receptors (MC3R and MC4R) in the central nervous system (CNS).The peptide binds to MC3R and MC4R receptors in the hypothalamus, a region of the brain that regulates sexual arousal and motivation. This activation stimulates neurological pathways that control sexual desire and arousal, leading to an increase in sexual motivation.

PT-141 OVERVIEW
Category: Cyclic Heptapeptide, Selective Melanocortin Agonist
How It Works: PT-141 (Bremelanotide) selectively activates Melanocortin Receptors (MC3R and MC4R) in the central nervous system (CNS). The peptide binds to MC3R and MC4R receptors in the hypothalamus, a region of the brain that regulates sexual arousal and motivation. This activation stimulates neurological pathways that control sexual desire and arousal, leading to an increase in sexual motivation.
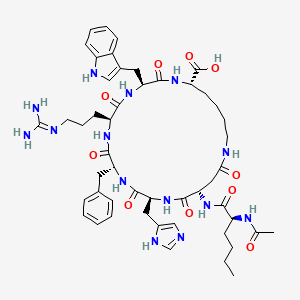
Chemical Structure: Ac-Nlee-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-OH
Alternative Names: Bremelanotide
CAS Number: 409740-19-4
WHAT IS PT-141
PT-141, officially named Bremelanotide, is a cyclic heptapeptide derived from the Melanotan family. It was developed as a synthetic peptide and a derivative of Melanotan 2, a tanning agent first studied in the 1980s.
-
Refinement: Unlike its parent compound, Melanotan 2, PT-141 was refined specifically for its effects on sexual desire and arousal, minimizing the undesirable tanning side effects.
-
Mechanism: It selectively activates Melanocortin Receptors (MC3R and MC4R) in the central nervous system (CNS) to stimulate neurological pathways that control sexual desire and arousal.
-
Regulatory Status: It is distinguished as the first FDA-approved treatment for female HSDD (Hypoactive Sexual Desire Disorder) that operates centrally, targeting the neurological components of sexual desire rather than the peripheral vascular systems.
WHAT DOES PT-141 DO?
PT-141 acts as an agonist, primarily targeting Melanocortin Receptors (MCRs) within the central nervous system (CNS). Unlike PDE-5 inhibitors such as Viagra, which act on blood flow, PT-141 targets the brain’s arousal pathways, making it one of the only therapies that directly enhances desire rather than just physical function.
Mechanism of Central Action
-
Central Desire Enhancement (MC4R): PT-141 shows a high affinity for MC3R and MC4R, which are abundantly expressed in the hypothalamus (EX: Paraventricular Nucleus). This region is a key center for regulating sexual behavior. Activation of these receptors is thought to stimulate dopamine and oxytocin release in reward pathways, thereby enhancing sexual motivation and desire.
-
Central Erection Response: In men, PT-141 initiates an erectile response via a CNS mechanism, triggering local nitric oxide (NO) release in penile tissues. This is independent of the peripheral vascular action of PDE5 inhibitors (EX: Viagra).
Pharmacokinetics
-
Half-Life: Following subcutaneous injection, PT-141 achieves peak plasma concentration in about 60 minutes. It has a short elimination half-life of approximately 2 to 3 hours.
-
Duration of Effect: Despite its short elimination half-life, clinical pharmacodynamic effects can last up to 16 hours.
BENEFITS/ CLINICAL TRIALS
PT-141 is unique because it goes beyond performance enhancement—it directly influences desire and motivation by targeting the brain's arousal pathways. Both clinical trials and anecdotal reports highlight its ability to improve sexual health in ways that traditional drugs cannot.
Central Mechanism of Action
PT-141 selectively activates Melanocortin Receptors (MC3R and MC4R) in the hypothalamus, a region of the brain that regulates sexual arousal and motivation.
-
Boosts Dopamine Signaling: By stimulating these receptors, PT-141 enhances dopamine activity, which plays a critical role in sexual drive and reward pathways.
-
Central Nervous System Focus: Because it works in the brain rather than the vascular system, PT-141 influences psychological arousal as well as physical readiness.
-
Independent of Nitric Oxide: Unlike traditional Erectile Dysfunction (ED) drugs (like PDE-5 inhibitors), PT-141 does not rely on the nitric oxide pathway. This mechanism makes it effective for individuals who don't respond to those inhibitors.
Key Benefits Observed in Clinical Trials
PT-141's development history includes both male ED and female HSDD studies, culminating in a specific FDA approval.
For Women (HSDD - FDA Approved)
-
FDA-Approved Treatment: Clinical studies demonstrated that PT-141 significantly increased sexual desire scores and related distress in women diagnosed with Hypoactive Sexual Desire Disorder (HSDD).
-
Improved Satisfaction: Women reported higher levels of sexual fulfillment and responsiveness, often leading to a statistically significant increase in satisfying sexual events.
- Neurobiological Effect: By acting on the brain, PT-141 helps restore the psychological component of arousal, which PDE-5 inhibitors cannot address.
-
Alternative for ED Non-Responders: Men who fail to respond to Viagra or Cialis often benefit from PT-141 since it bypasses nitric oxide pathways.
-
Enhanced Response: Early trials showed it was a potent initiator of penile erection in men with various forms of ED, including those unresponsive to PDE-5 inhibitors.
-
Boosts Desire: PT-141 is considered a "true" aphrodisiac, boosting the motivational and desire component of sexual behavior and enhancing both erection quality and sexual desire. It also showed synergistic effects when combined with low-dose Sildenafil.
SIDE EFFECTS
Like most peptides and medications, PT-141 carries some potential side effects. Side effects are common but generally mild-to-moderate and transient, typically occurring within a few hours of administration.
Most Common Side Effects
The most frequent reactions are linked to central nervous system activation and mild systemic changes:
-
Nausea (Most Frequent): Reported in up to 40 percent of participants in clinical studies. Nausea is dose-dependent, can be severe, and typically subsides within 2 hours of administration. Vomiting may also occur.
-
Headache and Flushing: Headache and facial flushing are commonly reported, caused by CNS activation and mild vascular changes.
-
Fatigue and Drowsiness: Temporary tiredness or somnolence may be experienced.
-
Injection Site Reactions: With subcutaneous administration, local reactions like redness, itching, swelling, or irritation at the injection site may occur.
Cardiovascular and Dermal Effects (Cautions)
-
Transient Blood Pressure Changes: PT-141 can cause a temporary, reversible rise in systolic and diastolic blood pressure (approximately 2 to 4 mmHg), peaking 4 to 8 hours after injection, along with a drop in heart rate.
-
Caution: Individuals with uncontrolled hypertension or pre-existing cardiovascular conditions should exercise caution and discuss these risks with a physician.
-
-
Skin Pigmentation Changes: Some users report darkening of existing moles (hyperpigmentation) or areas of the skin and gums (gingival hyperpigmentation). This side effect is linked to the peptide’s origin as a Melanocortin peptide derived from Melanotan II.
-
Note: In clinical trials, about 1 percent of patients reported dermal changes, and this effect may not be reversible in half of the affected patients.
-
IS PT-141 SAFE?
PT-141 (Bremelanotide) represents a new frontier in sexual health therapies. It is recognized as a game-changer in the future of sexual health by targeting the psychological and neurological aspects of arousal.
Unique Mechanism and Benefits
Unlike PDE-5 inhibitors such as Viagra, which only address blood flow, PT-141 works on the central nervous system, stimulating the brain’s arousal pathways to increase desire and motivation.
-
Targeted Desire: By targeting the psychological and neurological aspects of arousal, PT-141 goes beyond performance enhancement to address the root of desire itself.
-
For Women (Clinical Use): It is an FDA-approved treatment for Hypoactive Sexual Desire Disorder (HSDD), with strong clinical data backing its effectiveness.
-
For Men (Alternative Use): It offers a promising alternative for those who don't respond to traditional Erectile Dysfunction (ED) medications, improving both libido and performance.
-
Wellness Use: It has gained attention for its ability to enhance intimacy, connection, and overall sexual satisfaction.
Regulatory Status and Safety Takeaway
-
FDA Status: PT-141 (Bremelanotide) is an FDA-approved prescription drug for a specific indication (HSDD in premenopausal women).
-
Established Safety: When used under medical supervision at the prescribed dose of 1.75 mg SubQ, it has an established safety profile for short-term and 52-week use in premenopausal women.
-
Contraindications: It is contraindicated in patients with uncontrolled hypertension or cardiovascular disease due to its transient effects on blood pressure.
-
Banned Substance: The peptide is banned by WADA (World Anti-Doping Agency).
-
Long-Term Safety: Most clinical studies focus on short-term or on-demand use. Long-term effects are still being researched, so extended use should be approached carefully.
Overall, PT-141 is generally considered safe when used in controlled, as-needed doses, but users should monitor tolerance and consult with a healthcare provider, especially if they have underlying health conditions.
DOSAGE
Dosing is strictly regulated due to FDA approval for HSDD (Hypoactive Sexual Desire Disorder). PT-141 is generally used on an as-needed basis, not as a daily therapy.
Administration and Standard Therapeutic Dose
-
Administration: Self-administered subcutaneous (SubQ) injection into the abdomen or thigh.
-
Standard Therapeutic Dose (FDA Approved): 1.75 mg per injection.
-
Timing: Administered at least 45 minutes before anticipated sexual activity.
-
Typical Research/Off-Label Range: 250 mcg – 2 mg per dose, depending on individual sensitivity. Some individuals start with a lower dose (250–500 mcg) to test tolerance before increasing.
Frequency Restrictions
To minimize the risk of side effects and tolerance development, use of PT-141 is strictly limited:
-
Do not use more than one dose within 24 hours.
-
Do not use more than eight doses per month.
RECONSTITUTION
PT-141 (Bremelanotide) is typically supplied as a lyophilized (freeze-dried) powder in the research market, or as a pre-filled autoinjector in the pharmaceutical market.
Reconstitution and Dilution
-
Reconstitution Fluid: Use sterile or bacteriostatic water (BAC Water).
-
Mixing Technique: Inject the BAC Water slowly down the side of the vial. DO NOT SHAKE vigorously. Gently swirl the vial until the powder is completely dissolved into a clear solution.
-
Standard Dilution (EX: 10 mg Vial): For a 10 mg research vial, adding 2 mL of BAC water results in a concentration of 5 mg/mL.
-
Syringe Measurement: Adding 2 mL of BAC water means that 0.1 mL (10 units on an insulin syringe) contains 0.5 mg of PT-141.
Post-Reconstitution Storage
-
Storage: Store the vial in a refrigerator at 2–8°C. Do not freeze it again after reconstitution.
-
Stability: The solution should be used within 30 days to ensure optimal results and stability.
WHERE TO BUY PT-141
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for PT-141, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of PT-141 outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- “Bremelanotide.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/Bremelanotide. Accessed 24 Nov. 2025.
- by, Written. “PT-141 (Bremelanotide): Benefits, Dosage, Side Effects, and Safety.” Swolverine, swolverine.com/blogs/blog/pt-141-bremelanotide-benefits-dosage-side-effects-and-safety?_pos=1&_sid=a4ddff459&_ss=r. Accessed 24 Nov. 2025.
- “PT-141 for Men.” Tower Urology, 18 Sept. 2025, www.towerurology.com/mens-sexual-health/pt-141-for-men/.
- “How to Properly Mix Peptide PT-141 for Research Purposes.” UK Peptides - Buy High-Quality Research Peptides, www.uk-peptides.com/blog/how-to-mix-pt-141-for-research#:~:text=PT%2D141%20Peptide%20Vial%20(10mg,to%20500mcg%20of%20PT%2D141. Accessed 24 Nov. 2025.
-
The Journal of Clinical Endocrinology & Metabolism (PMC3380939). "$\text{Kisspeptin-}10$ Is a $\text{Potent}$ $\text{Stimulator}$ of $\text{LH}$ and $\text{Increases}$ $\text{Pulse}$ $\text{Frequency}$ in $\text{Men}$."
-
PubMed (ID 12851303). "$\text{PT}-141$: a $\text{melanocortin}$ $\text{agonist}$ $\text{for}$ $\text{the}$ $\text{treatment}$ of $\text{sexual}$ $\text{dysfunction}$."
-
PubMed Central (PMC6819021). "$\text{Bremelanotide}$ $\text{for}$ $\text{the}$ $\text{Treatment}$ of $\text{Hypoactive}$ $\text{Sexual}$ $\text{Desire}$ $\text{Disorder}$: $\text{Two}$ $\text{Randomized}$ $\text{Phase }3$ $\text{Trials}$."
-
Mayo Clinic. "$\text{Bremelanotide}$ ($\text{subcutaneous}$ $\text{route}$) - $\text{Side}$ $\text{effects}$ $\&$ $\text{dosage}$."
-
Concierge $\text{MD}$ $\text{LA}$. "$\text{PT}-141$ $\text{Side}$ $\text{Effects}$: $\text{Weighing}$ $\text{Costs}$ $\text{And}$ $\text{Benefits}$ $\text{Of}$ $\text{Use}$."
-
PubMed Central (PMC2694735). "$\text{Melanocortin}$ $\text{Receptors}, \text{Melanotropic}$ $\text{Peptides}$ $\text{and}$ $\text{Penile}$ $\text{Erection}$."
The PrepTide: Disclaimer

