TB-500
Helps to regulate the protein actin by binding to it, promoting cell migration, endothelial cell differentiation, and boosting the body's natural healing process.

TB-500 OVERVIEW
Category: Thymosin Beta-4 Fragment, Cell Migration promoter
How It Works: TB500 helps recruit the necessary cells to the site of injury, thereby enhancing the body’s natural healing processes.
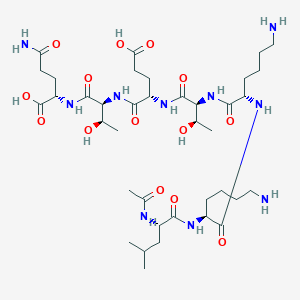
Chemical Structure: Ac-Ser-Asp-Lys-Pro-Asp-Met-Ala-Glu-Ile-Glu-Lys-Phe-Asp-Lys-Ser-Lys-Leu-Lys-Lys-Thr-Glu-Ser
Alternative Names: Thymosin Beta-4 Fragment, TB4 Fragment
CAS Number: 77591-33-4
WHAT IS TB-500
TB500, a synthetic version of thymosin beta-4, is gaining recognition for its extraordinary healing properties. This peptide is primarily known for its ability to accelerate tissue repair and regeneration, making it a valuable asset for those dealing with injuries or chronic conditions. The healing power of TB500 is facilitated by its capacity to promote cell migration and proliferation by representing the biologically active portion of the naturally occurring human protein TB4, which plays a major role inside of platelets and wound fluid, used for tissue repair, cell migration ,and blood vessel growth.
WHAT DOES TB-500 DO?
TB-500 is known for its systemic effects, meaning it acts throughout the body, unlike some peptides, which are more localized. Its mechanisms center on cellular organization and movement:
-
Actin Regulation: The primary molecular mechanism is the regulation of Actin, a protein essential for cell structure and movement. Tb4 fragments bind to Actin, promoting cell migration, proliferation, and differentiation processes critical for wound closure and tissue regeneration.
-
Angiogenesis Promotion: TB-500 stimulates the growth of new blood vessels, ensuring oxygen and nutrients are rapidly delivered to injured or ischemic tissues.
-
Anti-Fibrosis: Research suggests TB-500 exhibits anti-fibrotic activity, helping to reduce the formation of excessive scar tissue, leading to more functional and complete tissue repair.
-
Inflammation Modulation: It helps modulate the inflammatory response, reducing chronic or excessive inflammation, which can impair long-term healing.
When administered, TB500 helps recruit the necessary cells to the site of injury, thereby enhancing the body’s natural healing processes. This characteristic has positioned TB500 as a potent therapeutic agent for a variety of health issues.
BENEFITS/ CLINICAL TRIALS
While the full Tb4 protein has been investigated in clinical contexts, TB-500 itself remains a research compound
- Anti-Inflammatory Properties:
- In addition to tissue repair, TB-500 is also showing promise in anti-inflammatory properties. Inflammation can hinder recovery, and as such reduction of inflammation can reduce recovery times.
- Enhanced Vascularization Or Blood Flow:
- It has been linked to enhanced vascularization or the formation of new blood vessels.
- Athletic Performance
- Better exercise and recovery: For athletes and fitness enthusiasts, performance enhancement is a top priority. Tb-500 has been shown to be a promising tool to help in both energy levels and recovery times from workouts and physical activity.
- Improved mobility: It has also shown the ability to improve flexibility and mobility.
- Improved stamina: thanks to the enhanced blood flow rates, TB-500 helps boost oxygen delivery to muscles and can lead to prolonged performance during training and competitions.
Regulatory Status: No large-scale, independent Phase 3 human trials on TB-500 have established its clinical safety or efficacy for general use.
Scientific Research and Studies on TB500:
Medical and Athletic Interest
The scientific community has taken a keen interest in TB500 due to its potential applications in both medicine and athletics. Various studies have explored the peptide’s effects on tissue repair, inflammation reduction, and overall health enhancement. Research involving animal models has shown that TB500 can significantly improve wound healing rates and reduce the incidence of scars. For instance, studies demonstrated that when TB500 was applied to injured tissues, there was a notable increase in collagen deposition, a critical factor in tissue strength and integrity (U.S. National Library of Medicine, 2020).
Applications in Chronic Pain and Arthritis
In clinical settings, preliminary studies have also indicated that TB500 may have positive implications for patients with chronic pain conditions. Research has suggested that the peptide’s anti-inflammatory properties can lead to a reduction in pain levels, thereby improving the quality of life for individuals suffering from ailments such as arthritis. While these findings are promising, more extensive human clinical trials are required to establish definitive conclusions about TB500’s efficacy in treating chronic pain and other medical conditions.
Sports Medicine and Recovery
Moreover, the exploration of TB500 in the context of sports medicine has yielded encouraging results. Case studies involving athletes who utilized TB500 have shown significant improvements in recovery times and performance levels. These anecdotal reports, coupled with scientific investigations, illustrate the peptide’s potential as a powerful tool for enhancing physical capabilities. While the research is still in its early stages, the growing body of evidence surrounding TB500 continues to spark interest in its applications for healing and athletic performance.
SIDE EFFECTS
Due to its role as a naturally occurring signaling protein, TB-500 is generally considered low in toxicity in preclinical models. However, its unapproved status means the full risk profile in humans is unknown.
-
Systemic Effects:
-
Headache or dizziness (usually mild and transient).
-
Lethargy or tiredness (uncommon).
-
-
Local Reactions:
-
Injection Site Reactions: Pain, swelling, or redness at the subcutaneous injection site.
-
-
Unknown Risks: Because Tb4 promotes cell migration and proliferation, there is a theoretical risk regarding its potential interaction with pre-existing or latent malignancies (cancers), which warrants extreme caution and further research.
Legal and Anti-Doping Concerns
Furthermore, legality is another important aspect that users should consider. The regulatory status of TB500 varies by country, and it may be prohibited in competitive sports by organizations like the World Anti-Doping Agency (WADA). TB500 is listed under S2: Peptide Hormones, Growth Factors, and Related Substances, which makes its use banned for athletes in many sports. Athletes should verify the regulations surrounding TB500 in their respective sports to avoid potential disqualifications or penalties.
IS TB-500 SAFE?
TB-500 is not approved by the FDA or any major global regulatory authority for human therapeutic use.
While it is generally regarded as safe, thanks to the research conducted on it, it should be carefully considered based on its side effects and standing as a research-only peptide. If users have underlying health conditions, it should also be very carefully considered, as TB-500 has the potential to cause serious side effects.
Note: It has no regulations due to not being approved by the FDA, and as such, should also be looked at critically for which company you purchase it from.
DOSAGE
When considering the use of TB500, understanding the appropriate dosage and method of administration is crucial for maximizing its benefits.
The dosage can vary based on individual needs, the severity of the condition being treated, and the specific goals of the user.
For instance, typical dosages range from 2 to 5 mg per week, often administered in divided doses to maintain stable levels in the body. Some users may start with a loading phase, where higher doses are taken initially to saturate the system, followed by a maintenance dose.
RECONSTITUTION
TB-500 is supplied as a lyophilized (freeze-dried) powder and requires reconstitution with sterile water.
-
Reconstitution Fluid: Use sterile or bacteriostatic water.
- Draw Water: Using a sterile syringe (e.g., a 1 mL syringe), withdraw the desired amount of water (1 mL to 2 mL is standard).
- Inject Water: Slowly inject the water into the TB-500 vial. Aim the water stream at the side of the glass wall, not directly onto the powder, to ensure gentle dissolution.
- Mix Gently: Do not shake the vial vigorously. Gently swirl the vial or roll it between your hands until all the powder is completely dissolved and the solution is clear. This usually takes a few minutes.
- Storage: Once reconstituted, store the vial immediately in the refrigerator at 2-8°C (36-46°F). The solution is generally stable for up to 6 weeks when refrigerated and protected from light.
WHERE TO BUY TB-500
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for TB-500, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of TB-500 outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- Schreiner, M, et al. “A Primary Intravascular Synovial Sarcoma Causing Deep-Vein Thrombosis and Pulmonary Embolism in a 20-Year-Old Woman.” Current Oncology (Toronto, Ont.), U.S. National Library of Medicine, Oct. 2015, pmc.ncbi.nlm.nih.gov/articles/PMC4608415/.
- Maeda, Yoshiaki, et al. “Oleosome-Associated Protein of the Oleaginous Diatom Fistulifera Solaris Contains an Endoplasmic Reticulum-Targeting Signal Sequence.” Marine Drugs, U.S. National Library of Medicine, 30 June 2014, pmc.ncbi.nlm.nih.gov/articles/PMC4113804/.
- “PubMed.” National Center for Biotechnology Information, U.S. National Library of Medicine, pubmed.ncbi.nlm.nih.gov/. Accessed 21 Nov. 2025.
- Commissioner, Office of the. U.S. Food and Drug Administration, FDA, www.fda.gov/. Accessed 21 Nov. 2025.
- TB-500 Patient Information.Docx, happyhormonesmd.com/wp-content/uploads/2024/12/TB-500-Patient-Information.docx.pdf. Accessed 22 Nov. 2025.
- “TB 500 Trifluoroacetic Salt.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/TB-500-Trifluoroacetic-Salt. Accessed 21 Nov. 2025.
-
Journal of Regenerative Medicine. "Thymosin Beta-4 in the Regulation of Cell Migration, Angiogenesis, and Tissue Repair."
-
International Journal of Molecular Sciences. "The Role of Thymosin Beta 4 in Wound Healing and Fibrosis Reduction."
-
Wound Repair and Regeneration. "Thymosin beta 4 enhances dermal wound healing by regulating key cellular processes."
-
Regulatory Toxicology and Pharmacology. "Preclinical safety evaluation of Thymosin Beta-4: Implications for human use."
-
American Journal of Physiology—Heart and Circulatory Physiology. "Thymosin $\text{B}4$ promotes cardiac repair and regeneration after injury."
The PrepTide: Disclaimer

