VIP
VIP performs a wide array of functions, primarily involving smooth muscle relaxation, vasodilation, and the regulation of secretion and immune responses.
VIP OVERVIEW
Category: Neuropeptide, Neurotransmitter, Immunomodulator
How It Works: VIP binds to VPAC1 and VPAC2 receptors to act as a potent multi-system regulator. VIP acts as a potent vasodilator (widens blood vessels) and bronchodilator (relaxes lung airways), primarily involving smooth muscle relaxation. It acts as an anti-inflammatory agent, regulating T-cell cytokine balance to suppress excessive immune activity. It is involved in regulating secretion (glandular activity) and plays a key role in coordinating the body's circadian rhythms (sleep-wake cycles).
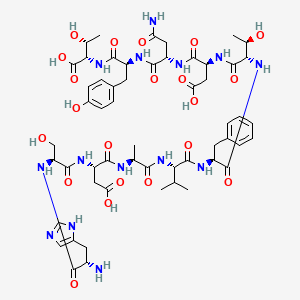
Chemical Structure: His-Ser-Asp-Ala-Val-Phe-Thr-Asp-Asn-Tyr-Thr-Arg-Leu-Arg-Lys-Gln-Met-Ala-Val-Lys-Lys-Tyr-Leu-Asn-Ser-Ile-Leu-Asn-NH2
Alternative Names: Vasoactive Intestinal Peptide
CAS Number: 38241-76-8
WHAT IS VIP
Vasoactive Intestinal Peptide (VIP) is a 28-amino-acid neuropeptide that belongs to the secretin-glucagon family of hormones. It performs a wide array of functions, primarily involving smooth muscle relaxation, vasodilation, and the regulation of secretion and immune responses.
Distribution and Role
VIP is widely distributed throughout the body, acting as a hormone in various peripheral organs and as a neurotransmitter/neuromodulator in the nervous system.
-
Nervous System Roles: It acts as a neurotransmitter in the CNS (Central Nervous System) and enteric nervous system (the gut's nervous system).
-
Peripheral Roles: It acts as a hormone/cytokine modulator in peripheral tissues, including the gut, lungs, heart, and pancreas.
WHAT DOES VIP DO?
VIP exerts its effects by binding to two high-affinity G-protein coupled receptors: VPAC1 and VPAC2. The receptor distribution explains its multifaceted actions across the entire body.
Actions Across Body Systems
-
Cardiovascular System: As its name implies, VIP is a potent vasodilator, helping to lower arterial blood pressure and increase blood flow in organs like the heart and lungs. It also has a positive effect on heart rate and contractility. VIP is one of the strongest known natural vasodilators.
-
Respiratory System: VIP causes smooth muscle relaxation in airways (bronchi), making it a key regulator of respiratory function (bronchodilation).
-
Digestive System: VIP relaxes smooth muscles in the gut, such as the lower esophageal sphincter, stomach, and gallbladder, aiding motility. It also stimulates the secretion of water, electrolytes, and bicarbonate into the intestines and pancreatic juice, while inhibiting gastric acid secretion.
-
Immune System: VIP has significant anti-inflammatory and immunomodulatory properties, helping to maintain immune homeostasis. It generally suppresses excessive Th1 and Th17 inflammation and promotes the development of regulatory T cells, promoting an anti-inflammatory state.
-
Central Nervous System (CNS): VIP functions as a neurotransmitter/neuromodulator, playing roles in the regulation of circadian rhythms, learning, memory, and anxiety. In the brain, VIP is highly concentrated in the Suprachiasmatic Nucleus (SCN), which is the body's master circadian clock, essential for synchronizing and regulating sleep-wake cycles and neuroendocrine function.
BENEFITS/ CLINICAL TRIALS
VIP has been investigated for conditions stemming from chronic inflammation, vasoconstriction, and immune dysregulation. It is a potent biological compound with wide-ranging systemic effects.
-
Pulmonary Arterial Hypertension (PAH): Its potent vasodilatory and bronchodilatory action in the lungs makes VIP a major therapeutic candidate for PAH (high blood pressure in the lungs). Clinical trials have focused on specialized delivery of VIP for PAH, often via inhalation
-
Chronic Inflammatory Response Syndrome (CIRS): VIP is actively researched for conditions like CIRS (often associated with Lyme disease or mold toxicity), where it aims to restore hypothalamic and immune function that may have been dysregulated by chronic inflammation.
-
Gastrointestinal and Autoimmune Conditions: Due to its Th1/Th17 suppressive effects, it is studied in models of IBD (Inflammatory Bowel Disease) and rheumatoid arthritis. EX: Treatment with VIP reduced the clinical and histopathologic severity of colitis in animal models
- Neuroprotection and Development: A large body of studies has associated VIP with neuroprotection. This includes observations that VIP expression is increased at the time of synapse formation and demonstrations of VIP neuroprotection against electrical blockade. Superactive neuroprotective lipophilic VIP analogs have also been synthesized.
-
Hormonal Responses: VIP enhanced cortisol secretion from human adrenal cells in a dose-dependent manner. Furthermore, in these cells, VIP significantly stimulated the release of dehydroepiandrosterone, testosterone, androstenedione, and aldosterone. VIP acted directly on adrenocortical carcinoma cell lines to stimulate steroidogenesis and cortisol secretion.
-
Cardiovascular System: Original studies identified increased VIP synthesis in the hypertensive rat model, suggesting a compensatory mechanism for VIP in the hypertensive state. VIP is 50–100 times more potent than acetylcholine as a vasodilator. VIP immunoreactive nerve fibers are present in key cardiac structures (sinoatrial node, atrium, interatrial septum, etc.).
- Diabetes: The VIP content of the gastroduodenal segment has been shown to be significantly lower in diabetic rats than in normal rats, suggesting a link to the progression of diabetes-related gut issues.
Cancer Research and Diagnostics
-
Cancer Receptor Expression: A majority of human tumors, especially the frequently occurring carcinomas (breast, prostate, colon, lung, and bladder), express VIP receptors (predominantly VPAC1 receptors). This suggests a role for VIP as a regulator of cell mitosis
-
Tumor Targeting: Radiolabeled peptides, such as 123I-VIP, have been used in peptide receptor scintigraphy to identify and localize tumors (like rare VIPomas and colorectal/gastric/pancreatic carcinoma) in a noninvasive manner before surgery and during follow up.
Clinical Trial Findings
-
Clinical trials have primarily focused on specialized delivery of VIP for PAH (often via inhalation) and small Phase 1/2 studies in neurological and autoimmune diseases, confirming its transient vasodilatory effects and overall tolerability.
SIDE EFFECTS
Side effects of VIP are typically mild and often related to its core pharmacodynamic action as a potent vasodilator (widening of blood vessels). These effects are generally more pronounced with higher or more rapid administration.
Common Systemic Effects
-
Flushing/Redness: A temporary sensation of warmth or redness (facial flushing) in the face or body is the most common side effect.
-
Neurological/Cardiovascular: Headache (due to vasodilation) and transient hypotension (low blood pressure) are common.
-
Dizziness/Lightheadedness: Changes in blood pressure can cause dizziness, especially if standing up too quickly after administration.
Other Reported Effects
-
Gastrointestinal/CNS: Mild nausea or dizziness.
-
Injection Site Reactions: Minor pain, swelling, or irritation may occur at the injection site.
-
Urinary: VIP may have a slight diuretic effect, leading to increased urination.
Inhalation Side Effects
For specialized administration routes, side effects can include:
-
Respiratory: Dry mouth or throat, cough, or mild bronchoconstriction (rare).
IS VIP SAFE?
VIP is an unapproved research peptide without FDA certification, and its exogenous, high-dose use is restricted.
Safety Profile and Tolerability
-
Naturally Occurring: VIP is a naturally occurring peptide hormone in the body, which contributes to its generally favorable safety profile.
-
Clinical Studies: Clinical studies often describe VIP as well-tolerated, with a low incidence of severe adverse effects in appropriate doses.
-
Potential for Interactions: Like any medication, VIP can interact with other drugs, so it is important to inform your healthcare provider of all medications and supplements you are taking.
Safety and Administration Takeaway
-
Professional Supervision is Key: Safety relies heavily on administration under the guidance of a knowledgeable physician. They can ensure the correct dosage and administration method are used for the specific condition being treated, minimizing risks.
-
Dosing Management: Safety relies heavily on expert dosing and administration protocol, particularly to manage the highly predictable vasodilatory effects (flushing, headache) and avoid hypotensive episodes (low blood pressure).
-
Usage Restriction: Its use is strictly reserved for controlled research.
DOSAGE
Dosing is complex and highly dependent on the administration route (systemic vs. pulmonary targeting).
Administration and Half-Life
-
Half-Life Constraint: VIP has an extremely short plasma half-life of approximately 2 minutes, necessitating infusion or specialized nasal/pulmonary delivery for sustained effect.
-
Administration Routes: Typically administered via Intravenous (IV) infusion, Subcutaneous (SubQ) injection, or Inhalation/Nebulization (the preferred route for pulmonary targeting).
-
Injection Site: Administer injections into subcutaneous fat (such as the stomach, thigh, or arm).
Example Dosing and Cycling
-
Dosing Range (Clinical Research): Usually administered in the microgram range (EX: 50 mcg to 200 mcg per dose) or via continuous infusion.
-
Injections Per Week: 5 injections weekly (for systemic effect).
-
Time of Day: VIP can be injected any time of day. It is typically administered in the morning or early afternoon to align with its natural role in regulating circadian rhythms, energy levels, and cellular function.
-
Recommended Cycles: 3 months on, 1 month off. 3 cycles per year.
Dilution Example
-
Vial Details: 5 mg VIP / 5 mL bacteriostatic water.
-
Concentration: 1 mg/mL (1,000 mcg/mL).
-
Duration: Approximately 5 weeks (25 doses).
RECONSTITUTION
VIP is supplied as a lyophilized (freeze-dried) powder. VIP is highly unstable once mixed, requiring immediate use and specific storage.
Reconstitution Fluid and Dilution
-
Reconstitution Fluid: Use sterile or bacteriostatic water (BAC Water) or saline.
-
Standard Dilution (EX: 10 mg Vial): For a 10 mg vial, adding 1 mL of BAC water results in a highly concentrated stock solution of 10 mg/mL.
-
Alternative Dilution (EX): Adding 2 mL of water to a 10 mg vial yields a concentration of 5 mg/mL.
-
Mixing Technique: Inject the diluent gently and swirl until dissolved. Do not shake vigorously.
Post-Reconstitution Storage (CRITICAL)
-
Storage: The solution must be stored in the refrigerator (2°C to 8°C).
-
Stability Warning: Once reconstituted, the solution is highly unstable and must be used within 24 hours to minimize degradation and ensure potency.
WHERE TO BUY VIP
Researchers should always vet their sources to ensure that a few key factors are present in their test subjects. With the rise in peptide popularity in recent years, many companies have created peptides that undergo little to no testing, quality standards, or regulations. As it is not regulated by the FDA, researchers must do their due diligence and look closely at the company's practices and standards.
When selecting a supplier for VIP, focus on transparency and quality assurance, not customer testimonials:
- Quality Documentation: A reputable supplier must provide:
- Certificate of Analysis (COA): This document must be recent (corresponding to the batch/lot number purchased) and demonstrate a minimum purity of >95% via High-Performance Liquid Chromatography (HPLC) testing.
- Mass Spectrometry (MS) Data: The COA must include mass spectrometry (MS) confirmation to verify the compound’s exact molecular weight, confirming its chemical identity.
- Contaminant Testing: Look for reports on heavy metals, microbial load, and solvent residues (e.g., residual trifluoroacetic acid, or TFA). The presence of these contaminants can severely compromise research and introduce unknown toxicity.
- Vendor Verification and Transparency
-
Specialization: Prioritize vendors who specialize in the manufacturing and distribution of peptides for academic and biotechnology research, rather than general supplement vendors.
-
Manufacturing Origin: Inquire about the source of the raw materials and the manufacturing protocols. Ideal suppliers adhere to strict quality control processes.
-
Handling & Storage: The supplier must provide clear documentation on the proper storage and handling procedures for the peptide to maintain its stability and integrity.
-
Conclusion on Procurement: Given the high risk of contamination, mislabeling, and legal ambiguity. The use of VIP outside of this defined research context poses unacceptable, unquantified risks to human health.
REFERENCES
- Iwasaki, Mari, et al. “Recent Advances in Vasoactive Intestinal Peptide Physiology and Pathophysiology: Focus on the Gastrointestinal System.” F1000Research, U.S. National Library of Medicine, 12 Sept. 2019, pmc.ncbi.nlm.nih.gov/articles/PMC6743256/#:~:text=Vasoactive%20intestinal%20peptide%20(VIP)%20is,barrier%20function%2C%20and%20mucosal%20immunology.
- Potential Clinical Applications of Vasoactive Intestinal Peptide: A Selected Update - Sciencedirect, www.sciencedirect.com/science/article/abs/pii/S1521690X04000570. Accessed 26 Nov. 2025.
- “Vasoactive Intestinal Peptide.” National Center for Biotechnology Information. PubChem Compound Database, U.S. National Library of Medicine, pubchem.ncbi.nlm.nih.gov/compound/53314964. Accessed 26 Nov. 2025.
- Ganea, D, et al. “The Neuropeptide Vasoactive Intestinal Peptide: Direct Effects on Immune Cells and Involvement in Inflammatory and Autoimmune Diseases.” Acta Physiologica (Oxford, England), U.S. National Library of Medicine, Feb. 2015, pmc.ncbi.nlm.nih.gov/articles/PMC4484298/#:~:text=In%20addition%2C%20systemic%20administration%20of,et%20al.%2C%201983).
- Dr Miles Nichols & Nicola Schuler. “Peptide Therapy: Vasoactive Intestinal Polypeptide (VIP).” Medicine With Heart, 1 Feb. 2023, medicinewithheart.com/vasoactive-intestinal-polypeptide-vip/#:~:text=Despite%20all%20of%20the%20benefits,with%20VIP%20or%20other%20peptides.
-
Paragon $\text{Sports}$ $\text{Medicine}$. "$\text{SS-31}$ $\text{Peptide}$ $\text{Cellular}$ $\text{Energy}$ $\&$ $\text{Repair}$."
-
PubMed (ID 32273339). "$\text{The}$ $\text{mitochondria-targeted}$ $\text{peptide}$ $\text{SS-31}$ $\text{binds}$ $\text{lipid}$ $\text{bilayers}$ $\text{and}$ $\text{modulates}$ $\text{surface}$ $\text{electrostatics}$ $\text{as}$ $\text{a}$ $\text{key}$ $\text{component}$ of $\text{its}$ $\text{mechanism}$ of $\text{action}$."
-
Frontiers $\text{in}$ $\text{Immunology}$. "$\text{The}$ $\text{Role}$ of $\text{Kisspeptin}$ in $\text{the}$ $\text{Control}$ of $\text{the}$ $\text{Hypothalamic-Pituitary-Gonadal}$ $\text{Axis}$ $\text{and}$ $\text{Reproduction}$."
-
PubMed (ID 12851303). "$\text{PT}-141$: a $\text{melanocortin}$ $\text{agonist}$ $\text{for}$ $\text{the}$ $\text{treatment}$ of $\text{sexual}$ $\text{dysfunction}$."
-
ResearchGate (ID 49800137). "$\text{Tesamorelin}$."
-
Actas $\text{Dermo-Sifiliográficas}$. "$\text{Melanotan}$ $\text{II}$ $\text{and}$ $\text{Its}$ $\text{Clinical}$ $\text{Implications}$."
-
MDPI (ID 1422-0067/23/19/11991). "$\text{MOTS-c}, \text{the}$ $\text{Most}$ $\text{Recent}$ $\text{Mitochondrial}$ $\text{Derived}$ $\text{Peptide}$ in $\text{Human}$ $\text{Aging}$ $\text{and}$ $\text{Age-Related}$ $\text{Diseases}$."
-
The PrepTide: Disclaimer

